US Drug Watchdog Now Invites Top Personal Injury Law Firms To Join Then In A State Specific Effort To Identify And Help More Victims Of A Failed All Metal Hip Implant
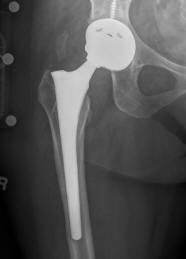
(PRWEB) October 16, 2013 -- The US Drug Watchdog says,"After working with recipients of failed metal-on-metal hip implants for years, we are convinced the true number of all-metal hip implant failure victims who are still unidentified is in the hundreds of thousands nationwide. We believe, for instance, there might be hundreds, if not thousands of DePuy pinnacle all-metal hip implant victims in almost every state.
To change this reality, the US Drug Watchdog is now inviting highly-regarded personal injury law firms from every state to participate with them in a customized state-specific approach. This invitation to collaborate with the group is to identify more victims of a failed metal-on-metal hip implant. The bottom line is to remove all possibility that even one single victim will be left behind with the financial burden of a very costly revision surgery. For more information, personal injury law firms that are well established, have a superior intake, and case management capabilities in each state are urged to contact the US Drug Watchdog at 866-714-6466. http://USDrugWatchdog.Com
One of the biggest problems with the metal-on-metal hip implants failures is the lack of an aggressive national campaign to get the word out to actual victims. There is also the lack of well publicized information about the additional serious issues associated with a failed all-metal hip implant, such as possible blood poisoning related to elevated levels of cobalt and chromium in the recipient's blood stream. Further, the US Drug Watchdog believes the vast majority of metal-on-metal hip implant failure victims want to keep their case with a highly-regarded local personal injury law firm in their state, as opposed to a national law firm, or a marketing law firm.
The US Drug Watchdog's state specific initiative for well established personal injury law firms includes: 20+ targeted press releases branding the participating local law firm as the go to place for a recipient of a DePuy Pinnacle or other metal-on-metal hip implants that are subject to national litigation. The group's initiative also includes the possibility of time on a local TV channel or in the local news to increase public awareness about metal-on-metal hip implant failure issues and other potentially serious medical issues associated with all-metal hip implants. The campaign more than pays for itself with one new client.
The US Drug Watchdog is strongly encouraging managing partners, or senior-level partners who share their passion about helping metal-on-metal hip implant recipients with a failed device, to call them at 866-714-6466, about this creative and aggressive initiative. http://USDrugWatchdog.Com
Metal on Metal hip implant manufacturers that are now subject to national litigation over the issue of premature failures:
- DePuy Pinnacle Case Number U.S. District Court for the Northern District of Texas MDL#2244
- DePuy ASR Case Number Ohio Southern Federal District Court Case Number MDL No. 2197AAJ
- Stryker Rejuvenate Hip Implant Stem Failure Teleseminar: Metallosis with No Metal on Metal Articulation will address the issues surrounding the latest hip replacement systems recall. On July 6, 2012 Stryker issued a voluntary recall of its Rejuvenate and ABG II modular-neck stems in the United States
- Biomet M2a Magnum Metal-on-Metal Hip Replacement U.S. District Court for the Northern District of California. (Case No. 3:12-cv-3476) filed on July 2nd 2012
- Zimmer Durom Cup Product Liability mass tort litigation, MDL NO. 2158 (SDW), which is pending in the federal District Court in New Jersey (time frame 2006-July 2008)
- Wright Medical Technology, Inc., Conserve Hip Implant Products Liability Litigation ("MDL No. 2329"). This is now pending in the U.S. District Court for the Northern District of Georgia.
For attribution purposes please review the following information about all metal hip implant failures:
For attribution please review: Med Page Today: May 17th 2013 http://www.medpagetoday.com/Orthopedics/Orthopedics/39234
For more information about a MRI and tissue inflammation with a metal on metal hip implant we have attached a link to the May 15th 2013 Science Daily news article: http://www.sciencedaily.com/releases/2013/05/130515151436.htm
For more information about the recalled DePuy ASR, or the DePuy Pinnacle all metal hip implant, please review the The New York Times article dated May 16th 2013. http://www.nytimes.com/2013/05/17/business/jj-is-phasing-out-metal-replacement-hips.html
M Thomas Martin, Americas Watchdog, http://chinesedrywallcomplaintcenter.com/, 866-714-6466, [email protected]



Share this article