Corporate Whistleblower Center Urges Medical Device Or Pharmaceutical Industry Insiders To Call Them About Reward Programs For Defective Products
(PRWEB) November 14, 2013 -- The Corporate Whistleblower Center says, "According to provisions of the False Claims Act, whistleblowers receive a portion of the settlement as a reward for their information, usually between 10 and 30 percent. We are extremely interested in talking to pharmaceutical companies or medical device insiders if they possess well documented proof a current drug or medical device is defective, because the rewards really can be in the millions." http://CorporateWhistleblowerCenter.Com
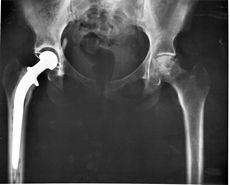
In one recent example, medical device company Boston Scientific Corp and its subsidiaries Guidant LLC, Guidant Sales LLC, and Cardiac Pacemakers Inc agreed to pay $30 million to settle allegations that it violated the False Claims Act*. According to a press release from the Department of Justice, the company allegedly sold defective implantable defibrillators. The heart devices were then implanted into Medicare patients.
In this case, the whistleblower will receive $2.25 million as a reward for providing the incriminating evidence.
The Corporate Whistleblower Center says, "If you are a manager, sales rep, or a insider at a drug or medical device company, and you have solid proof your products are hurting people, please call us at 866-714-6466, so we can help you not only do the right thing, but also try to get you rewarded for your information." http://CorporateWhistleblowerCenter.Com
Simple rules for a whistleblower from the Corporate Whistleblower Center:
- Do not go to the government first if you are a major whistleblower. The Corporate Whistleblower Center says, “Major whistleblowers frequently go to the federal government thinking they will help. It’s a huge mistake.”
- Do not go to the news media with your whistleblower information. Public revelation of a whistleblower’s information could destroy any prospect for a reward.
- Do not try to force a government contractor, or corporation to come clean to the government about their wrongdoing. The Corporate Whistleblower Center says, “Fraud is so rampant among federal contractors that any suggestion of exposure might result in an instant job termination, or harassment of the whistleblower. Come to us first, tell us what type of information you have, and if we think it’s sufficient, we will help find the right law firms to assist in advancing your information.”
- Do not submit information to law firms online. The Internet is loaded with phony websites asking whistleblowers to send in their information to unknown law firms. The Corporate Whistle Blower Center warns, “Do not send anything through a website. If you want to know who the best whistleblower law firms are, we will tell you; once we know what type of information you possess, your level of proof, and the nature of the scheme.”
- The Corporate Whistleblower Center wants to emphasize there are high quality whistleblowers in every state including California, New York, Florida, Texas, Massachusetts, Maryland, Virginia, Ohio, Pennsylvania, West Virginia, Tennessee, North Carolina, Georgia, Alabama, Louisiana, Missouri, Michigan, Iowa, Indiana, Illinois, Wisconsin, Minnesota, North Dakota, Nebraska, Kansas, Colorado, Utah, New Mexico, Arizona, Nevada, Idaho, Oregon, Washington, and Alaska.
Any type of insider, or employee, who possesses significant proof of their employer or a government contractor bribing foreign officials is encouraged to contact to Corporate Whistle Blower Center anytime at 866-714-6466, or they can contact the group via their web site at http://CorporateWhistleBlowerCenter.com.
*For attribution purposes, please review the October 17th 2013 news article on this topic http://www.corporatecrimereporter.com/news/200/bostonscientificfalseclaims10172013/
Case Number: The relevant case citation is U.S. ex rel. Allen v. Guidant Corp., No. 11-22 , D. Minn.
M Thomas Martin, America's Watchdog 6, http://CorporateWhistleblowerCenter.Com, +1 (866) 714-6466, [email protected]


Share this article