ReliantHeart HeartAssist5® VAD achieves CE Mark for European distribution
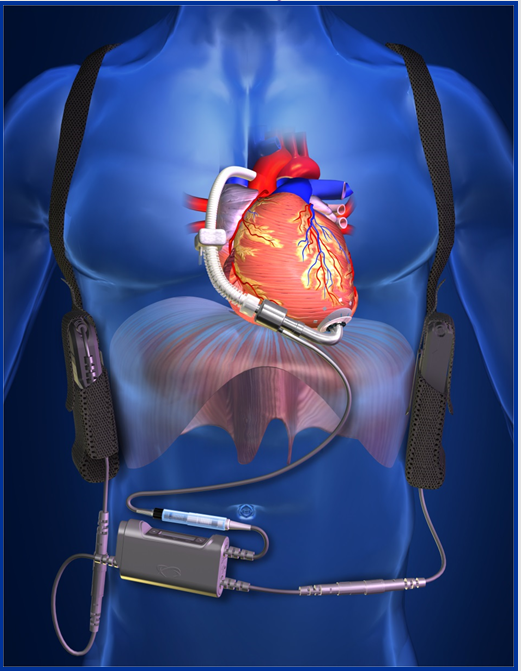
HOUSTON, TX (PRWEB) November 10, 2013 -- The ReliantHeart HeartAssist 5®Ventricular Assist Device (VAD) has been CE Mark* approved in Europe for use in patients requiring ventricular support due to end stage heart failure. The HeartAssist 5® is approved for bridge to transplant (BTT) patients awaiting cardiac transplantation and for destination therapy (DT) patients who are not candidates for cardiac transplantation.
The CE Mark for the HeartAssist 5® was previously granted to MicroMed Technology, Inc. and was first available for European distribution in 2009. Through completion of a court approved transaction, ReliantHeart, Inc. has become the successor-in-interest to the assets of MicroMed Technology, Inc., and will manufacture, sell and support the HeartAssist5® and its suite of VAD products and accessories.
An amendment to the earlier CE mark was granted October of 2012 to allow around the clock remote monitoring with the HeartAssist5® Conquest Controller™, a lightweight wearable controller designed to collect critical flow data. That data, as well as speed and power measurements, are transmitted to caregivers through the exclusive HeartAssist5® Remote Monitoring System. The HeartAssist5® is the only remotely monitored VAD in the world.
ReliantHeart competes in Europe with two companies: Thoratec (THOR) and HeartWare (HTWR). On November 6, 2013, HeartWare announced strong third quarter results of a 140% increase over same period last year. Revenue from international markets, generated through the sale of 287 units, was $26.6 million, an increase of approximately 38% from $19.3 million in the third quarter of 2012.
ReliantHeart intends to enter the U.S. market through an IDE (Investigational Device Exemption), which will allow the HeartAssist5® to be sold for use in a clinical trial approved by the FDA to collect safety and effectiveness data.
*The European Union applies the CE (Conformité Européenne) Marking process to ensure patient/user safety and to enforce that the manufacturer’s device fulfills what it is designed to perform.
Sailesh Saxena, ReliantHeart, Inc., http://www.reliantheart.com, 713-457-1480, [email protected]


Share this article