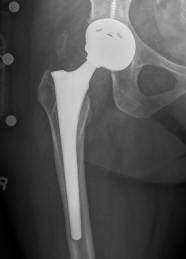
US Drug Watchdog Now Offers To Help All DePuy Pinnacle Hip Implant Recipients Get Identified If Their Hip Implant Has Failed With The Goal Being Compensation-Nationwide
(PRWEB) July 08, 2013 -- The US Drug Watchdog is calling their initiative DePuy Pinnacle metal on metal hip implant recipient initiative vital, because according to court records 130,000 US citizens are recipients, and only a fraction have been identified by the Texas federal court handling the litigation. The US Drug Watchdog says, "There has been very little news publicity about the DePuy Pinnacle metal on metal hip implant, and premature failures, and our big fear now is the time to get identified for what could be significant financial compensation will come, and go, because either the DePuy hip implant recipient never heard about premature hip implant failure issues, or they would rather live in pain than go through hip implant replacement surgery again.' In the hopes of dramatically changing the numbers of DePuy Pinnacle hip implant recipients who might have a premature failure the US Drug Watchdog is urging recipients to call them about a very simple blood test that measures cobalt, and chromium levels in a all metal hip implant recipients blood stream. If the cobalt,, and chromium levels are elevated it probably indicates a failure. For more information individuals who have a DePuy Pinnacle metal on metal hip implant are urged to contact the US Drug Watchdog at 866-714-6466.
The US Drug Watchdog says, "We frequently get asked what states were the DePuy Pinnacle all metal hip implant sold in, and our answer is all states including California, New York, New Jersey, North Carolina, Florida, Georgia, Texas, Louisiana, New Mexico, Massachusetts, Maine, Michigan, Minnesota, Missouri, Ohio, Oregon, Oklahoma, Nevada, Nebraska, Vermont, Virginia, Colorado, Connecticut, Idaho, Indiana, Illinois, West Virginia, and Washington." http://USDRugWatchdog.Com
The US Drug Watchdog indicates symptoms of the recalled DePuy ASR hip implant, or the DePuy Pinnacle failure include:
- Pain in the Hip Region
- Problems While Walking or The Inability to Walk
- Swelling of the Hip
- General Discomfort
- Lack of Flexibility
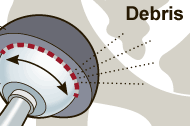
- Elevated levels of cobalt, or chromium in the hip implant recipients blood.
The US Drug Watchdog is the premier medical device, and pharmaceutical watchdog in the United States. The group says, “We want to make certain all DePuy Pinnacle hip implant recipients get a blood test to determine if their hip implant has failed. In the event the blood test indicates elevated levels of cobalt, and chromium we will get the recipient to national caliber law firms that have a superior record in representing their clients in these types of national lawsuits, involving potentially significant financial compensation." For more information about the DePuy Pinnacle metal on metal hip implant, and a critical blood test please contact the US Drug Watchdog anytime at 866-714-6466. http://USDrugWatchdog.com
U.S. District Court for the Northern District of Texas MDL#2244
Very Important Note From The US Drug Watchdog: The US Drug Watchdog says, "The statute of limitations on the recalled DePuy ASR metal on metal hip implant has not yet run out in the following states: Arkansas, Washington D.C., Florida, Maine, Maryland, Massachusetts, Michigan, Minnesota, Missouri, Montana, Nevada, New Hampshire, New Mexico, New York, North Carolina, North Dakota, Rhode Island, South Carolina, South Dakota, Vermont, Washington, Wisconsin, and Wyoming. If you are a recipient of a recalled DePuy ASR, and you have not yet been identified by the court please call us as soon as possible at 866-714-6466, if the procedure was done in one of these states." http://USDrugWatchdog.Com
DePuy ASR Case Number Ohio Southern Federal District Court Case Number MDL # 2197
M Thomas Martin, Americas Watchdog, 866-714-6466, [email protected]

Share this article