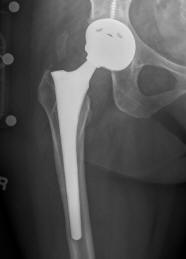
US Drug Watchdog Now Urges Recipients Of A Recalled DePuy ASR Metal Hip Implant To Get Identified Now If They Live In One Of The States Where There Is Still Time To Do So
(PRWEB) August 14, 2013 -- The US Drug Watchdog says, "We do not want one recipient of the recalled DePuy ASR all metal hip implant left holding the bag, with thousands of dollars in medical costs associated with a hip implant replacement surgery, also called a revision surgery. Unfortunately, there are less that 25 states remaining, where the statute he statute of limitations has not yet run out, for a recipient of a recalled DePuy ASR all metal hip implant, and we want to try to help every one of these recipients get identified to the court-before the allowable time to do so runs out. We need to emphasize we are talking about potentially hundreds of thousands of dollars in potential financial compensation, for each recipient." According to court records the statute of limitations for a recalled DePuy ASR metal on metal hip implant has not yet run out in Arkansas, Washington D.C., Florida, Maine, Maryland, Massachusetts, Michigan, Minnesota, Missouri, Montana, Nevada, New Hampshire, New Mexico, New York, North Carolina, North Dakota, Rhode Island, South Carolina, South Dakota, Vermont, Washington, Wisconsin, and Wyoming. The US Drug Watchdog is urging a recipient of the recalled DePuy ASR all metal hip implant, to call them immediately at 866-714-6466, if the surgical procedure to implant this type of hip implant was done in one of the states they have listed.
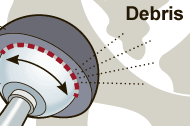
The US Drug Watchdog says, “We are also incredibly passionate about making certain all recipients of a DePuy Pinnacle metal on metal hip implant call us about a very simple blood test, that has been designed to measure for elevated levels of cobalt, and chromium in a recipients blood. According to records there are about 130,000 US citizens who have a DePuy Pinnacle all metal hip implant. That is enough people to fill to capacity the famed Rose Bowl in Pasadena, California, and Madison Square Gardens in New York City-combined. We would request recipients of this all metal hip implant device to call us at 866-714-6466, so we can explain the very simple process required for one of these blood tests, and the specific code for this blood test.” The DePuy Pinnacle metal on metal hip implant device is now the subject of a national class action lawsuit over issues related to premature failures. The focus of the US Drug Watchdog’s campaign is getting every recipient of the DePuy Pinnacle all metal hip implant identified, in the event their hip implant has failed, or the blood test indicates elevated levels of cobalt, and chromium in the recipients blood stream. According to the New York Times on December 27th 2011, “The so-called metal-on-metal hips, in which a device’s ball and joint are made of metal, are failing at high rates within a few years instead of lasting 15 years or more, as artificial joints normally do. The wear of metal parts against each other is generating debris that is damaging tissue and, in some cases, crippling patients.” http://USDrugWatchdog.Com
The US Drug Watchdog is the premier medical device, and pharmaceutical watchdog in the United States. The group says, “We want to every thing possible to assist recipients of the recalled DePuy ASR, and the DePuy Pinnacle, and individuals who have these specific types of all metal hip implants can call us anytime at 866-714-6466. http://USDrugWatchdog.com
U.S. District Court for the Northern District of Texas MDL#2244
DePuy ASR Case Number Ohio Southern Federal District Court Case Number MDL # 2197
The US Drug Watchdog says, "For more information please review the May 16th 2013 article in the New York Times, regarding the recalled DePuy ASR, and the DePuy Pinnacle all metal hip implants. http://www.nytimes.com/2013/05/17/business/jj-is-phasing-out-metal-replacement-hips.html
M Thomas Martin, Americas Watchdog, 866-714-6466, [email protected]

Share this article