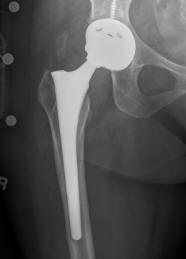
US Drug Watchdog Urges People With A Recalled DePuy ASR All Metal Hip Implant To Call Them ASAP If They Live In One Of The States Where Its Not Too Late To Get Signed Up
(PRWEB) August 16, 2013 -- The US Drug Watchdog says, "Unfortunately, for over twenty five states it is too late for us to identify recipients of recalled DePuy ASR all metal hip implant, because the statute of limitations has run out to get these individuals identified to the court handling the litigation related to this hip implant. However, if you had the surgical procedure done involving a recalled DePuy ASR hip implant in Arkansas, Washington D.C., Florida, Maine, Maryland, Massachusetts, Michigan, Minnesota, Missouri, Montana, Nevada, New Hampshire, New Mexico, New York, North Carolina, North Dakota, Rhode Island, South Carolina, South Dakota, Vermont, Washington, Wisconsin, or Wyoming we want to hear from you as soon as possible, because the statute of limitations has not yet run out in these states." If a recipient of a recalled DePuy ASR all metal hip implant has not yet been identified by the court handling the litigation related to this specific type of hip implant, and if the procedure was done in one of the states the group has listed, please call the US Drug Watchdog immediately at 866-714-6466."
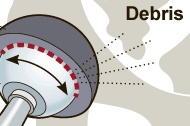
The US Drug Watchdog says, "If you are a recipient of a DePuy Pinnacle all metal hip implant, we are urging you to call us at 866-714-6466 for information related to a specific blood test that has been designed to detect elevated levels of cobalt, and chromium in a all metal hip implant recipient's blood. This is a big deal. If the cobalt, or chromium levels are elevated, we believe this is the best indicator for a all metal hip implant failure." A national lawsuit has been filed in federal court related to the DePuy Pinnacle metal on metal hip implants and premature failure issues. According to court records there are 130,000 US recipients of a DePuy Pinnacle all metal hip implant. The US Drug Watchdog is urging family members or friends of hip implant recipients who had their hip implant surgery between 2002 and 2011 to ask if they received a DePuy Pinnacle all metal hip implant. If the answer is yes, the US Drug Watchdog is urging the recipient or their family members to call them about this vital blood test. http://USDrugWatchdog.Com
The US Drug Watchdog is the premier medical device, and pharmaceutical watchdog in the United States. The group says, “We want to make certain all recalled DePuy ASR, or DePuy Pinnacle hip implant get help, and we are urging recipients of these specific type of hip implant devices to call us at 866-714-6466, so we can provide them with as much useful information at possible. We think it is particularly chilling that on May 16th 2013 the New York Times reported Johnson & Johnson's DePuy subsidiary is getting out of the all metal hip implant business." http://USDrugWatchdog.com
U.S. District Court for the Northern District of Texas MDL#2244
DePuy ASR Case Number Ohio Southern Federal District Court Case Number MDL # 2197
The US Drug Watchdog says, "For more information please review the May 16th 2013 article in the New York Times, regarding the recalled DePuy ASR, and the DePuy Pinnacle all metal hip implants. http://www.nytimes.com/2013/05/17/business/jj-is-phasing-out-metal-replacement-hips.html
M Thomas Martin, Americas Watchdog, 866-714-6466, [email protected]

Share this article