The ORLocate platform was demonstrated to the National Performance Advisory Groups(NPAG) HSDU and Operating Theatre Managers Forums at the Royal Asiatic Society in London
Swansea, West Glamorganshire (PRWEB UK) 7 August 2013 -- Haldor Advanced Technologies, a leading provider of RFID-enabled surgical instrument visibility solutions for the healthcare industry, was invited to demonstrate its offering to the NPAG HSDU and Theatres forums in London where there was significant interest in the traceability of surgical instruments.
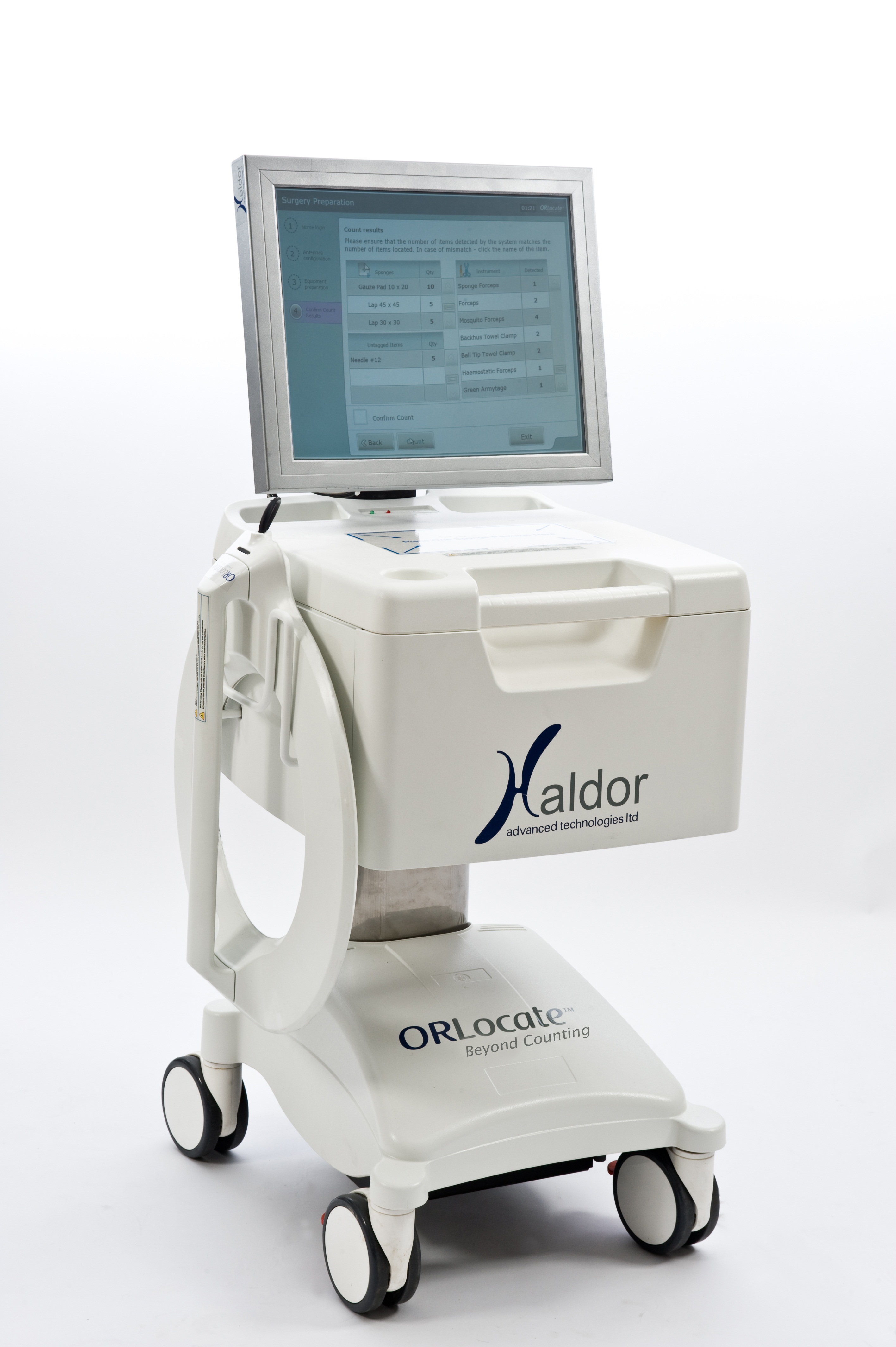
ORLocate® enables hospitals to significantly reduce costs associated to surgical instrument shrinkage as well as provide accurate usage data on each instrument to optimise instrument set size and overall inventory. The system helps increase staff productivity and accuracy by actively guiding and monitoring the technician’s entire workflow cycle to ensure compliance. It improves patient safety and infection control and prevents Retained Surgical Items (“RSI”) by provision of accurate instrument level data. Such data can also help the hospital to improve instrument utilisation; better accommodate their inventory to the real surgical needs while minimising redundancy, achieving better adjustment of the content of surgical sets. For more info, see a short video at the Haldor Technologies homepage.
The demonstration included a full cycle of surgical items from the HSDU to the Operating Theatre and back. After the demonstration, the forum discussed the recent video clip that was presented by Huw Edwards on the 10 o’clock BBC news on May 9th 2013 (http://www.bbc.co.uk/news/uk-22452328) referring to more than 762 “Never Events” in the last 4 years in UK hospitals. The Department of Health defines incidents such as operating on the wrong body part or leaving instruments inside patients, Retained Surgical instrument (“RSI”) as “Never Events” (http://www.bbc.co.uk/news/health-22466496). The BBC news clip showed a patient named Donna Bowett being interviewed by Nicola Beckford, that suffered for months from 7-inch forceps that were left in her abdomen after a gallbladder operation. Many of the attendees spoke about their own experiences with such events indicating that the numbers could in fact be higher than reported. It is not difficult to understand how these events cost the NHS Millions of pounds per year.
Paul Wilson, recently retired Director of NPAG, who has facilitated the group for the last 13 years commented that “the feedback from the Group regarding the OR locate solution was very positive. The increase in traceability and the reduction in staff time at the end of each procedure in counting and checking instruments is a clear benefit. The NPAG Theatres Group does not endorse any commercial product as it is up to each theatre management team to make their own assessments based on local knowledge and practice. However, the clear business case and very quick return on investment must make this solution worthy of a more detailed assessment.”
Alan Heath, UK sales director commented that “The OR Locate solution can eliminate Retained Surgical Instrument risks and track theatre instruments and sponges in theatres and beyond. Instruments can be tracked throughout their life cycle complying with NICE & MHRA guidelines re CJD. ORLocate is the only GS1-compliant and UDI-ready RFID-enabled solution in the UK market.”
The ORLocate modular platform can be tailored to meet a small budget set level tracking solution while retaining extensibility for future expansion such as intra-operative counting of instruments, instrument level tracking and tailored solutions for loaned sets, peel-packs and more.
About Haldor Advanced Technologies
Haldor Advanced Technologies is committed to helping hospitals meet the ongoing challenge of increasing operational efficiency, reducing costs, and improving patient safety by enabling hospitals to effectively track, manage, and analyze surgical instruments on an individual basis. Haldor’s leading surgical instrument visibility solution, ORLocate® empowers hospitals to significantly reduce costs associated to surgical instrument shrinkage in both the operating room and sterile processing departments, to increase staff productivity by eliminating manual counting procedures, and to improve patient safety by managing infection control and the prevention of retained surgical items with accurate instrument level data. Headquartered in Hod Hasharon, Israel, Haldor has offices in Europe, the Middle East, and North America. Haldor has received ISO 13485 certification for a medical device quality system and is also a GS1 industry partner. Haldor's technology is patent protected in many countries. The ORLocate system has FDA 510K clearance and CE certification and the product is GS1-compatible and UDI-ready.
For more information, please visit http://www.haldor-tech.com
Ram Alt, Haldor Advanced Technologies, http://www.haldor-tech.com, +972 54-2003333, [email protected]


Share this article