Xcelthera Inc Secures First U.S. Patent for Large-Scale Production of High Quality Human Embryonic Stem Cells and their Neuronal and Heart Muscle Cell Therapy Products
San Diego, CA (PRWEB) May 08, 2014 -- Xcelthera Inc, a major innovator in the stem cell research market and one of the first U.S. companies formed for clinical applications of human embryonic stem cell (human ES cell) therapeutic utility for unmet medical needs, and its joint research partner San Diego Regenerative Medicine Institute announced today that the U.S. Patent and Trademark Office (USPTO) has granted Patent No. 8,716,017 entitled, “Technologies, Methods, and Products of Small Molecule-Directed Tissue and Organ Regeneration from Human Pluripotent Stem Cells.” This newly-issued patent is the first among a portfolio of intellectual property of Xcelthera Inc covering PluriXcel human stem cell technology platform for large-scale production of high quality clinical-grade pluripotent human ES cell lines and their functional human neuronal and heart muscle cell therapy products.
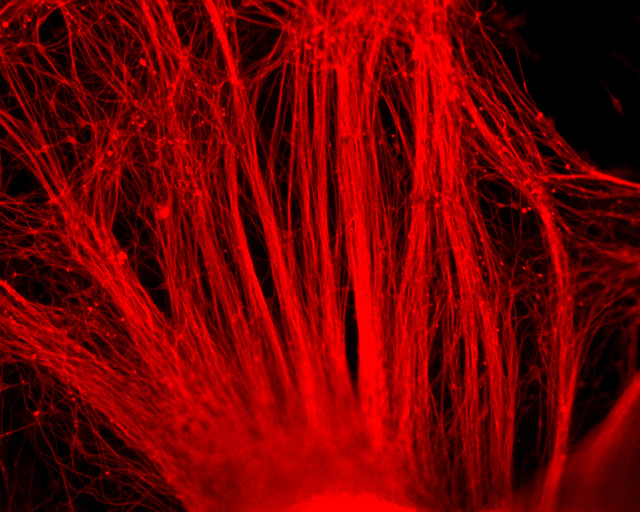
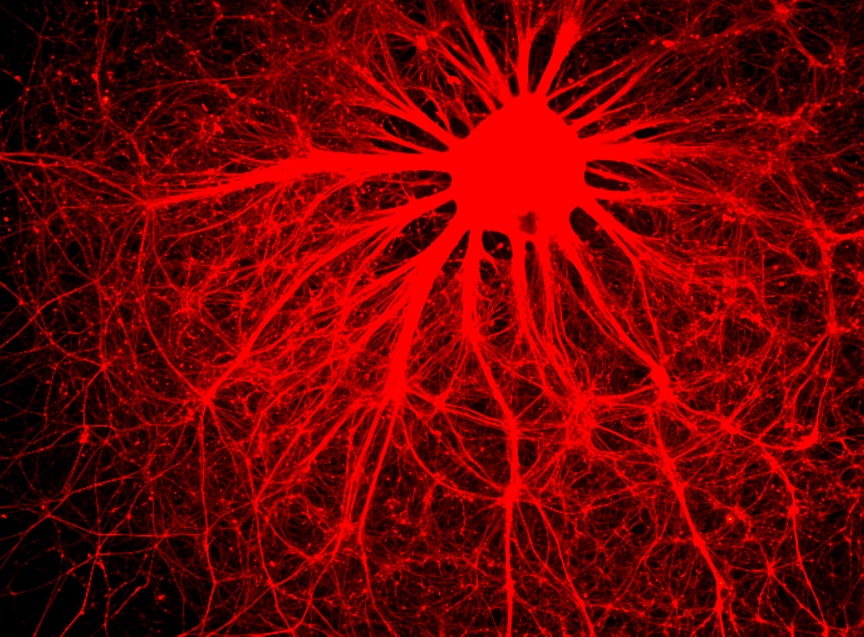
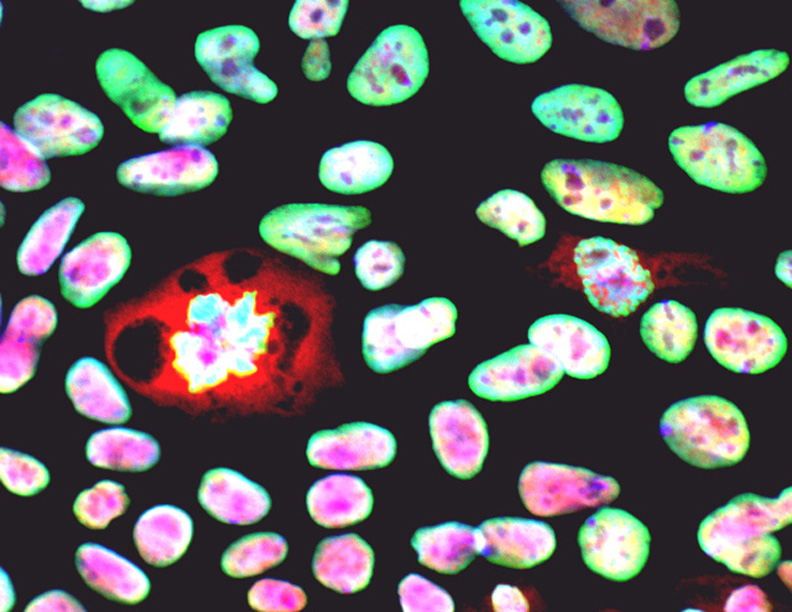
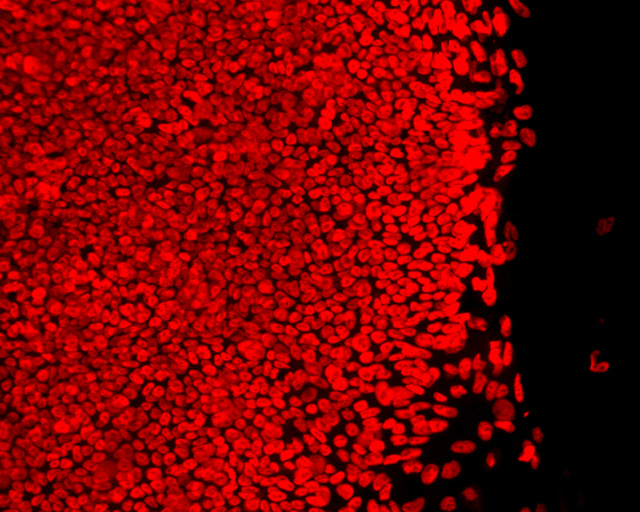
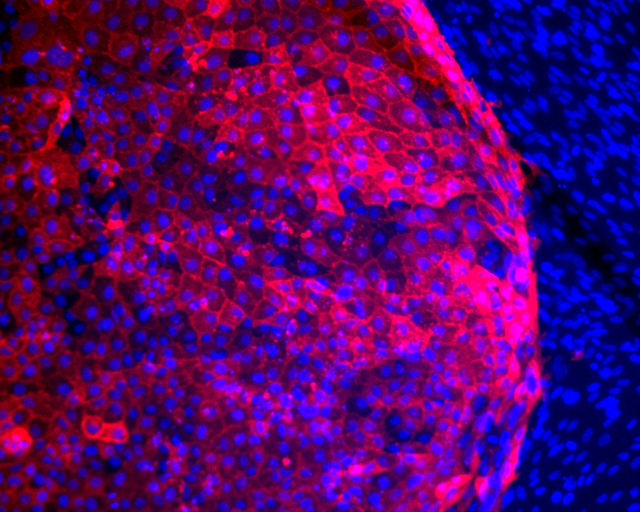
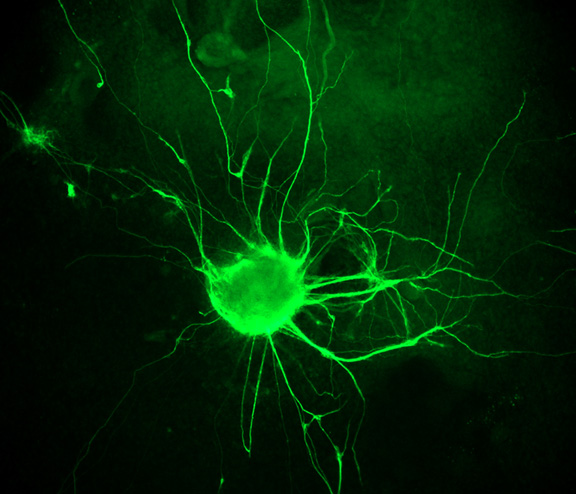
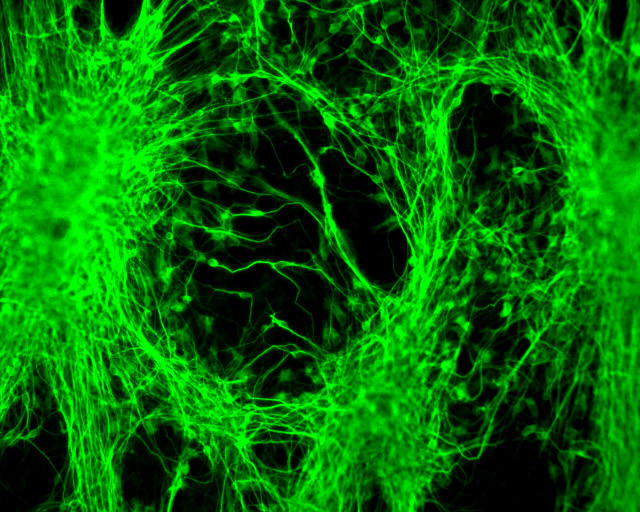
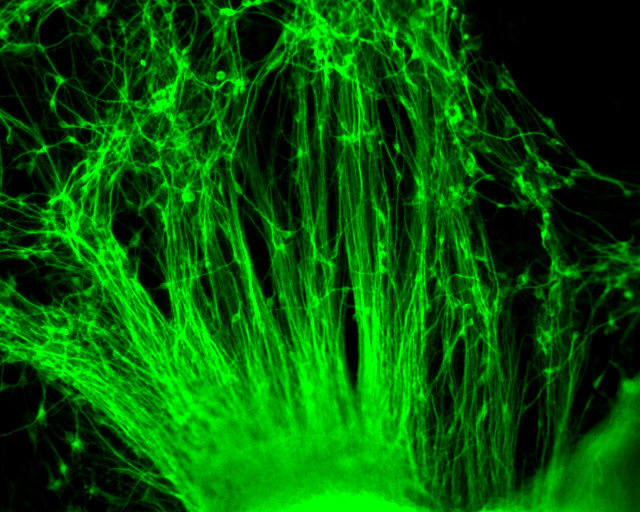
Neurodegenerative and heart diseases are major health problems and cost the worldwide healthcare system more than $500 billion annually. The limited capacity of these two cell systems -- neurons and cardiomyocytes -- for self-repair makes them suitable for stem cell-based neuronal and heart therapies. Nevertheless, to date, the existing markets lack a clinically-suitable human neuronal cell source or cardiomyocyte source with adequate regenerative potential, which has been the major setback in developing safe and effective cell-based therapies for neurodegenerative and heart diseases. Xcelthera proprietary PluriXcel technology allows efficient derivation of clinical-grade human ES cell lines and direct conversion of such pluripotent human ES cells by small molecule induction into a large commercial scale of high quality human neuronal or heart muscle cells, which constitutes clinically representative progress in both human neuronal and cardiac therapeutic products for treating neurodegenerative and heart diseases.
PluriXcel technology of Xcelthera Inc is milestone advancement in stem cell research, offering currently the only available human cell therapy products with the pharmacological capacity to regenerate human neurons and contractile heart muscles that allow restitution of function of the central nervous system (CNS) and heart in the clinic. Through technology license agreement with San Diego Regenerative Medicine Institute, Xcelthera Inc has become the first in the world to hold the proprietary breakthrough technology for large-scale production of high quality clinical-grade pluripotent human ES cell lines and their functional human neuronal and heart cell therapy products for commercial and therapeutic uses.
As neurodegenerative and heart diseases incur exorbitant costs on the healthcare system worldwide, there is a strong focus on providing newer and more efficient solutions for these therapeutic needs. Millions of people are pinning their hopes on stem cell research. PluriXcel technology platform of Xcelthera Inc is incomparable, providing life scientists and clinicians with novel and effective resources to address major health concerns. Such breakthrough stem cell technology has presented human ES cell therapy derivatives as a powerful pharmacologic agent of cellular entity for a wide range of incurable or hitherto untreatable neurodegenerative and heart diseases. Introduction of medical innovations and new business opportunities based on PluriXcel technology will shape the future of medicine by providing pluripotent human ES cell-based technology for human tissue and function restoration, and bringing new therapeutics into the market.
About Xcelthera Inc.
Xcelthera INC (http://www.xcelthera.com) is a new biopharmaceutical company moving towards clinical development stage of novel and most advanced stem cell therapy for a wide range of neurological and cardiovascular diseases with leading technology and ground-breaking medical innovation in cell-based regenerative medicine. The Company was recently incorporated in the state of California to commercialize the technologies and products developed, in part, with supports by government grants to the founder, by San Diego Regenerative Medicine Institute (SDRMI), an non-profit 501C3 tax-exempt status independent biomedical research institute that is interested in licensing its PATENT RIGHTS in a manner that will benefit the public by facilitating the distribution of useful products and the utilization of new processes, but is without capacity to commercially develop, manufacture, and distribute any such products or processes. Xcelthera is a major innovator in the stem cell research market and one of the first companies formed for clinical applications of human embryonic stem cell (human ES cell) therapeutic utility for unmet medical needs. The Company is the first to hold the proprietary breakthrough technology for large-scale production of high quality clinical-grade pluripotent human ES cell lines and their functional human neuronal and heart muscle cell therapy products for commercial and therapeutic uses. The Company owns or has exclusive rights in a portfolio of intellectual property or license rights related to its novel PluriXcel human stem cell technology platforms and Xcel prototypes of human stem cell therapy products. The inception of Xcelthera is driven by the urgent need for clinical translation of human ES cell research discoveries and innovations to address unmet medical challenges in major health problems. Xcelthera breakthrough developments in human ES cell research dramatically increase the overall turnover of investments in biomedical sciences to optimal treatment options for a wide range of human diseases. The overall strategy of the Company is to use cutting-edge human stem cell technology to develop clinical-grade functional human neural and cardiac cell therapy products from pluripotent human ES cells as cellular medicine or cellular drugs to provide the next generation of cell-based therapeutic solutions for unmet medical needs in world-wide major health problems. The Company is currently offering Series A Convertible Preferred Stock to accredited investors through equity crowdfunding to raise fund for its pre-IPO business operation and filing confidential IPO as an emerging growth company according to the JOBS Act to create a public market for its common stock and to facilitate its future access to the public equity market and growth of the Company.
Visit Xcelthera Inc. at http://www.xcelthera.com.
For more information or investment opportunity about Xcelthera series A round, please contact:
Xuejun H Parsons, PhD,
Chief Executive Officer
Xcelthera Inc.
http://www.xcelthera.com
888-706-5396 or 858-243-2046
investors(at)xcelthera.com or parsons(at)xcelthera.com
About San Diego Regenerative Medicine Institute
San Diego Regenerative Medicine Institute (http://www.sdrmi.org), a nonprofit independent biomedical research institute with 501C3 IRS tax-exempt status, was founded in 2010 to facilitate the transition of human stem cell research towards stem cell therapy to provide the next generation of cell-based therapeutic solutions for unmet medical challenges in world-wide major health problems. SDRMI is committed to the leading position in pluripotent human ES cell research and cell-based regenerative medicine by developing groundbreaking human ES cell technology platforms and innovative cell-based regenerative medicine approaches.
Visit San Diego Regenerative Medicine Institute at http://www.sdrmi.org.
For more information, please contact: contacts(at)sdrmi.org
Xuejun Parsons, Xcelthera Inc, http://www.xcelthera.com, +1 888-706-5396, [email protected]
Share this article