Psychiatric Drugs Send 90,000 to Emergency Rooms Each Year—Mental Health Watchdog Cites Need for Consumers to Search Psychiatric Drug Database for Documented Risks
Los Angeles, California (PRWEB) September 08, 2014 -- A recent JAMA Psychiatry study revealed that between 2009 and 2011, there were an estimated 267,000 emergency room visits because of adverse drug events from psychiatric drugs such as overdose, excessive sleepiness and head injuries. Over 19% of these emergency room visits resulted in patient hospitalization.[1] The lead author of the JAMA study suggested the need for caution, increased surveillance, and reduced use of psychiatric drugs, and even encouraged alternative treatments.[2]
The JAMA report, first released in July, breaks down the adverse drug events by class of drug, reporting that nearly 78,000 of the emergency room visits were due just to sedatives/anxiolytics, antidepressants and antipsychotics. Other drugs causing such reactions included lithium salts and stimulants.[3]
Citizens Commission on Human Rights (CCHR), the leading mental health watchdog, says this data should be of no surprise given the ever-increasing use of psychiatric drugs:
• According to IMS Health’s Total Patient Tracker database, psychiatric drug use now includes even the youngest of the nation's population. In fact, in 2013, more than one million American children between the ages of 0 and 5 were prescribed psychiatric drugs.[4]
• For all ages, more than ten million Americans took ADHD drugs, 41 million took antidepressants, more than six and a half million took antipsychotics and 36 million took anti-anxiety drugs.[4]
• Seventy-nine million Americans, one-in-four, are currently prescribed psychiatric drugs.[4]
In an effort to provide accurate information about the documented risks associated with psychiatric drugs, CCHR last year launched a comprehensive website and enhanced psychiatric drug database.
This database could not have been more timely or user friendly. According to the Food and Drug Administration (FDA), between 2004 and 2012, there were more than five hundred thousand adverse reactions from psychiatric drugs reported to the federal agency.[5] These reports are just the tip of the iceberg, as the FDA admits that only about 1% of serious adverse events are actually reported.[6]
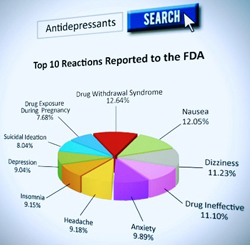
More telling, however, is that most Americans are unaware of the national and international regulatory agency warnings on most psychiatric drugs. For example, there are 134 drug regulatory warnings associated with antidepressants, 44 regulatory warnings for ADHD drugs and 25 regulatory warnings for anti-anxiety drugs.[7]
CCHR's website and drug database provides a wealth of information for families, including adverse side effects, the number of studies showing adverse effects of specific drugs and the FDA's data on adverse drug events. In short, the mental health watchdog has taken very complex data from a number of sources and provided an easy, user-friendly, format not available from the FDA or any other regulatory agency.
While many of the psychiatric drugs long have been branded with the FDA's "black box" warning—the federal agency's most serious warning—too often the adverse reactions associated with psychiatric drugs are down played and physician follow-up is spotty, leaving those taking psychiatric drugs to find accurate information on their own.
CCHR says that, “the JAMA study reinforces the need for full disclosure and, with 79 million Americans taking at least one psychiatric drug, the study's limited data at least provides an honest appraisal of the growing problem of adverse events associated with psychiatric drugs.”
For those concerned about the dangers associated with psychiatric drugs, the following links provide a complete list of all documented side effects of the following categories of psychiatric drugs:
ADHD Drug Side Effects
Antidepressant Drug Side Effects
Antipsychotic Drug Side Effects
Anti-anxiety Drug Side Effects
Read full article here.
About Citizens Commission on Human Rights: CCHR is a non-profit, non-political, non-religious mental health watchdog. Its mission is to eradicate abuses committed under the guise of mental health and enact patient and consumer protections. CCHR has helped to enact more than 150 laws protecting individuals from abusive or coercive mental health practices.
--
[1] Lindsey Tanner, "STUDY: PSYCH DRUG ER TRIPS APPROACH 90,000 A YEAR," Associated Press, July 9, 2014, bigstory.ap.org/article/study-psych-drug-er-trips-approach-90000-year-0.
[2] Lindsey Tanner, "STUDY: PSYCH DRUG ER TRIPS APPROACH 90,000 A YEAR," Associated Press, July 9, 2014, bigstory.ap.org/article/study-psych-drug-er-trips-approach-90000-year-0; Sabriya Rice, “Nearly 90,000 ED visits annually linked to psychiatric drug events,” Modern Healthcare, 3 Sept. 2014, modernhealthcare.com/article/20140903/NEWS/309039964.
[3] Lee M. Hampton, MD, MSc, et al., “Emergency Department Visits by Adults for Psychiatric Medication Adverse Events,” JAMA Psychiatry, 9 July 2014, ncbi.nlm.nih.gov/pubmed/25006837.
[4] IMS, Vector One: National (VONA) and Total Patient Tracker (TPT) Database, Year 2013, Extracted June 2014.
[5] "The Adverse Event Reporting System (AERS): Older Quarterly Data Files," U.S. Food and Drug Administration, fda.gov/Drugs/GuidanceComplianceRegulatoryInformation/Surveillance/AdverseDrugEffects/ucm083765.htm.
[6] David A. Kessler, MD, “Introducing MEDWatch A New Approach to Reporting Medication and Device Adverse Effects and Product Problems,” JAMA, 2 June 1993, Vol. 269, No. 21, pp. 2765-2768, fda.gov/downloads/Safety/MedWatch/UCM201419.pdf.
[7] International warnings compiled from the US FDA, fda.gov/Safety/MedWatch; Health Canada, hc-sc.gc.ca/index-eng.php; Japan Ministry of Health, Labour and Welfare, mhlw.go.jp/english/; Australia’s Therapeutic Goods Administration, tga.gov.au; UK’s Medicines and Healthcare products Regulatory Agency, mhra.gov.uk/; New Zealand's Medsafe, medsafe.govt.nz/; Irish Medicines Board, imb.ie/; South Africa's Medicines Control Council, sanctr.gov.za/YourRights/TheMedicinesControlCouncil/tabid/176/Default.aspx.
Media Department, Citizens Commission on Human Rights, http://www.cchrint.org, +1 (323) 467-4242, [email protected]
Share this article