Si14 Introduces the Revolutionary D-EYE Smartphone Retinal Screening System
Padova, Italy; Boston, MA, USA (PRWEB) October 06, 2014 -- Si14, an original design and technology development laboratory, introduced today the innovative D-EYE smartphone retinal screening system, a phone-case-sized add-on that turns an iPhone or Android smartphone into a fundus camera capable of taking high-definition video and still images of the eye for health screening and evaluation. The new smartphone lens system will be demonstrated at the D-EYE booth (#2275) at the American Academy of Ophthalmology annual meeting, Oct. 18-21, McCormick Place, Chicago, IL.
“With the compact D-EYE fundoscope camera lens, we are transforming the smartphone from a popular device for snapping ‘selfies’ to a valuable health examination tool that can aid in the discovery and diagnosis of eye diseases and abnormalities. The easy-to-use D-EYE affords examiners a clear view of both the anterior and posterior segments of the eye, allowing qualified medical professionals to examine, diagnose and grade a variety of diseases, ranging from diabetic retinopathy to glaucoma to cataracts and more,” said Si14 CEO Andrea Tellatin.
D-EYE is the result of ground-breaking work done by Dr. Andrea Russo, who partnered with Si14 in the development of the device. An ophthalmologist and Ph.D.-candidate researcher with the University of Brescia, Italy, Dr. Russo invented the D-EYE to improve access to vital medical screenings for people in need.
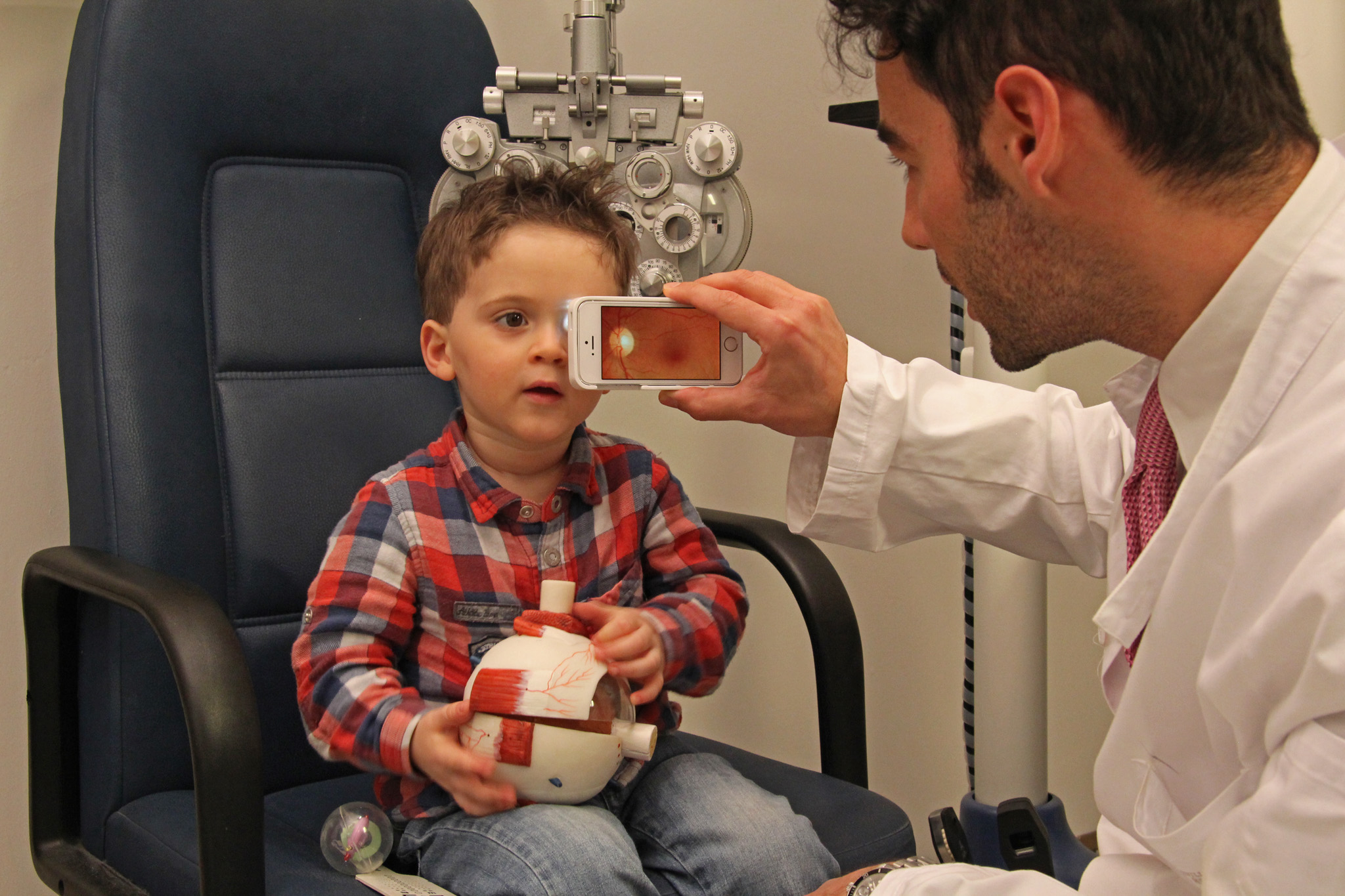
The D-EYE retinal screening system represents a novel, effective and low-cost approach to retinal screening, and it is the only truly portable ophthalmoscope capable of digitally imaging the fundus of the eye – small enough to go anywhere and requiring no external power source. The D-EYE device simply attaches to a smartphone (Apple iOS or Android), positioned over the phone’s camera lens and LED light source, enabling the phone to capture high-definition video and still images of the fundus of the eye. A D-EYE software app installed on the smartphone provides step-by-step instructions and archives all examinations on a real clinical chart for convenient file management for each patient.
Using the D-EYE attachment on the mobile device, the examiner can see the fundus of the eye from a non-invasive distance, and record and post-process the recorded frames. The images then can be sent wirelessly to the secure cloud-based patient medical record, where appropriately trained medical specialists in any location can review the images and form the proper diagnosis.
According to Dr. Russo, “The D-EYE retinal screening system can be used by a variety of health professionals ranging from ophthalmologists, neurologists, general practitioners, emergency physicians and pediatricians to school nurses, EMTs, and others. The system offers a quick, accurate and inexpensive way to examine the human eye and identify a variety of health conditions.”
The D-EYE smartphone retinal screening system offers:
• Convenient and ergonomic assessment of bedridden patients as well as children, even neonatal patients, whose spontaneous reaction to the light permits the device to capture retinal images
• Easy viewing of the optic nerve head (even without dilating eye drops) for detecting glaucoma
• Cataract diagnosis and grading
• Age-related macular degeneration (AMD) screening
• Diabetic retinopathy screening and grading
• Hypertensive retinopathy screening and grading
• Detection of neurological disorders, hemorrhages, arteriolar constriction, blood vessel abnormalities, maculopathy, cotton wool spots, exudates, neuritis, otticopatia and other diseases and conditions
• Visual acuity testing for adults and children
“Exams can be conducted in doctors’ offices, remote health clinics, hospitals, school nurse stations, urgent care centers, pharmacies and other locations, opening up a broad selection of examination points, enabling wide-spread screening in developing countries and bringing critical eye screening services to people in even the most remote locations,” Dr. Russo said.
The patent-pending D-EYE device is currently in clinical trial in support of Class II approval by the US Food and Drug Administration, and undergoing certification in Italy (recognized throughout the European Union), CE compliance, and UL approval in the United Kingdom. Physicians interested in participating in clinical evaluations to test the validity and operational characteristics of the D-EYE system should contact the company at [email protected].
About Si14
Si14 is a visionary original design and development laboratory for novel technology products that enhance the way we live, work and play. Founded in 2008 with offices in Padova, Italy, and Boston, MA, USA, Si14 turns inspiration into the realization of compelling, cutting-edge products and services that help people connect with their activities and their world, empower enterprises in realizing greater efficiencies, improve access to vital health screening services for millions worldwide, and much more. An Official Supplier to the Luna Rossa America’s Cup Team seeking the sailing world’s most coveted prize, Si14 enjoys partnerships with many leading companies and organizations across the technology spectrum, providing Si14 clients with unique insight, experience and access to the latest advances in embedded systems, bio and motion sensing, mobile communications and more. For more information on the D-EYE smartphone retinal screening system, please visit http://www.d-eyecare.com or call 978.224.1350 (US).
# # #
JIM WHITE, jameswhite9060, http://www.d-eyecare.com, +1 6268403796, [email protected]

Share this article