PANDA Realizes Continued Process Verification in Pharmaceutical Manufacturing
Freiburg, Germany (PRWEB) March 19, 2015 -- The EMA and FDA demand high transparency, verifiability at any time and complete documentation of the life cycle of every single pharmaceutical product. The manufacturing process must be permanently under control and any procedural deviations have to be recognized at the very moment they occur. The procedure allowing this is called Continued Process Verification by the FDA and Continuous Process Verification by the EMA.
The implementation of the CPV represents an enormous challenge for the pharmaceutical industry, whose details and standards in many respects still have to be worked out. Already after the first drafts of the new FDA Guidance on Process Validation in 2008, a forward-thinking pharmaceutical company started the development of a database system with the automated integration of different source systems and statistical software with which valuable experience was gained. This development work led to the now mature system PANDA.
THE REQUIREMENTS OF CONTINUED PROCESS VERIFICATION
The implementation of the Continued Process Verification demands the continuous collection and processing of the data of diverse parameters, such as the Critical Quality Attributes (CQA) and the Critical Process Parameters (CPP) in periodic data analyses.
Continuous reporting to the authorities is required, which can be achieved through quality and process reports (similar to the Annual Process Report) at regular intervals (e.g. monthly). In the data analyses, methods of statistical analysis such as trending, control charts or CpK (Process Capability Index) calculations should therefore be used where possible. The manual and product-oriented data transfer from LIMS systems and process data systems such as EBR (Electronic Batch Records) or BDE (“Betriebsdatenerfassung”/ production data acquisition) and their aggregation (e.g. on Excel sheets) are no longer suitable for Continuous Process Verification due to the increased amount of data and complexity.
In addition to minimizing risk and enormous cost reduction, the database-driven system for process analysis offers the further advantage that its scope goes far beyond the creation of regular and comprehensive reports. Once the data is in a structured form with dedicated access options, it can be used for complex purposes (such as Process Improvement).
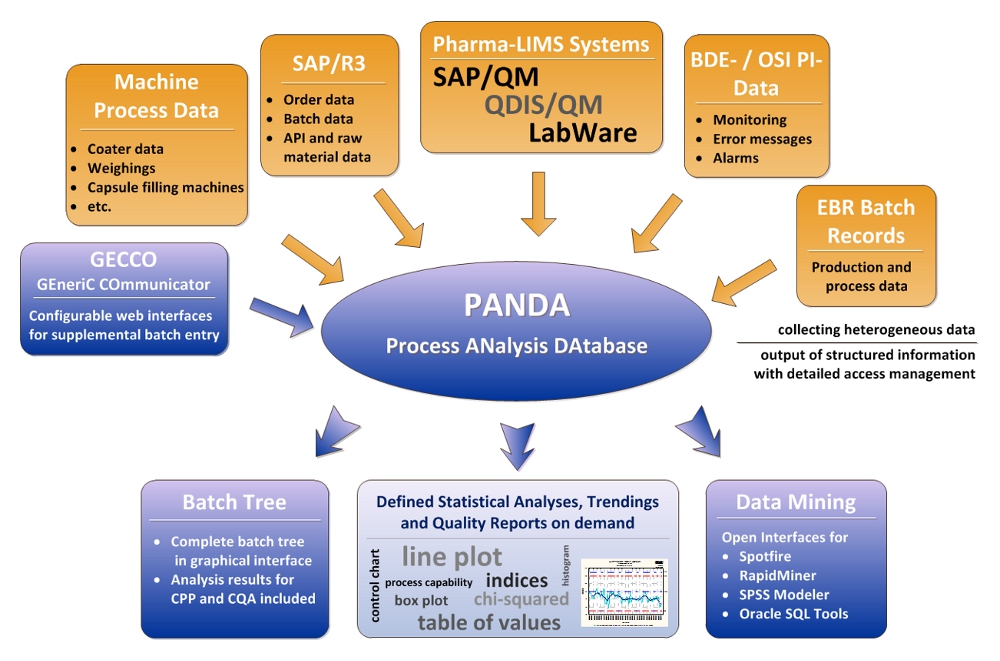
PANDA PROVIDES THE SOLUTION FOR THE CPV
In order to realize the Continued Process Verification and its complex tasks, Atris Information Systems – in close collaboration with Boehringer Ingelheim – has developed the standardized system PANDA.
PANDA is an integrated database system, processing and providing information from different source systems. With PANDA raw material, drug data, development data, process data and machine data as well as ERP data can be networked, structured and aggregated. PANDA allows the monitoring of CQA and CPP using trending and statistical analyses over long periods of time.
THE CYCLIC PANDA PROCESS
The PANDA process consists of five steps. In a first step, the different raw material, laboratory and machine process data with the associated metadata is imported into the system under the fullest satisfaction of all requirements for data integrity. Subsequently, a harmonization of the data takes place. This central function of the system only creates the conditions for being able to correlate the individual parameters. The user of the system then has the possibility to define data grids and parameters and thus obtain individual evaluations. In a next step the aggregation and analysis of the data is carried out.
At the end of the process, the results can be presented by using pattern reports. The data is kept automatically up to date by the import cycle, whereas reports can be accessed in an actualized version at any time.
PANDA PRIVATE CLOUD
The PANDA private cloud service adds another feature with numerous user advantages. In addition to the now widely known benefits of the private cloud, the company offers advisory services for statistical processing and know-how in the GMP environment. The requirements of the FDA and EMA for dealing with CPP and CQA data can be realized perspicuously in this way. The costs of the installation, validation and maintenance can be reduced by keeping full back-up service and data security and benefiting from synergy effects. In a professional environment at the technological forefront, Big Data Management and Process Verification become reliable, traceable and safe.
CONCLUSION
With PANDA, the result of the practical experience and development in recent years, a fully deployable solution for Continued Process Verification is available. PANDA offers numerous options for individual configurations by the user in terms of parameters, the evaluations and reports, and thereby represents a surplus for each pharmaceutical company.
ABOUT ATRIS:
Atris Information Systems GmbH is specialized in quality-assuring data processing and evaluation (business intelligence, trending, and data mining) in the pharmaceutical and chemical industry as well as in project and system validation in the GMP domain (research, production and quality assurance).
----------------------------------------
Footnote:
1 Guideline on process validation for finished products – information and data to be provided in regulatory submissions – February 27, 2014
http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2014/02/WC500162136.pdf
2 Guidance for Industry – Process validation: general principles and practices – January 2011
http://www.fda.gov/downloads/Drugs/Guidances/UCM070336.pdf
3 See also http://www.atris-systems.com/172.0.html
Peter Trochim, Atris Information Systems GmbH, http://www.atris-systems.com, +49 761 47 99 5920, [email protected]
Share this article