North American Surgeon is First in Virginia to use SpineFrontier’s Arena-C HA, a New Spinal Implant Manufactured with Innovative New Material to Enhance Spinal Fusion.
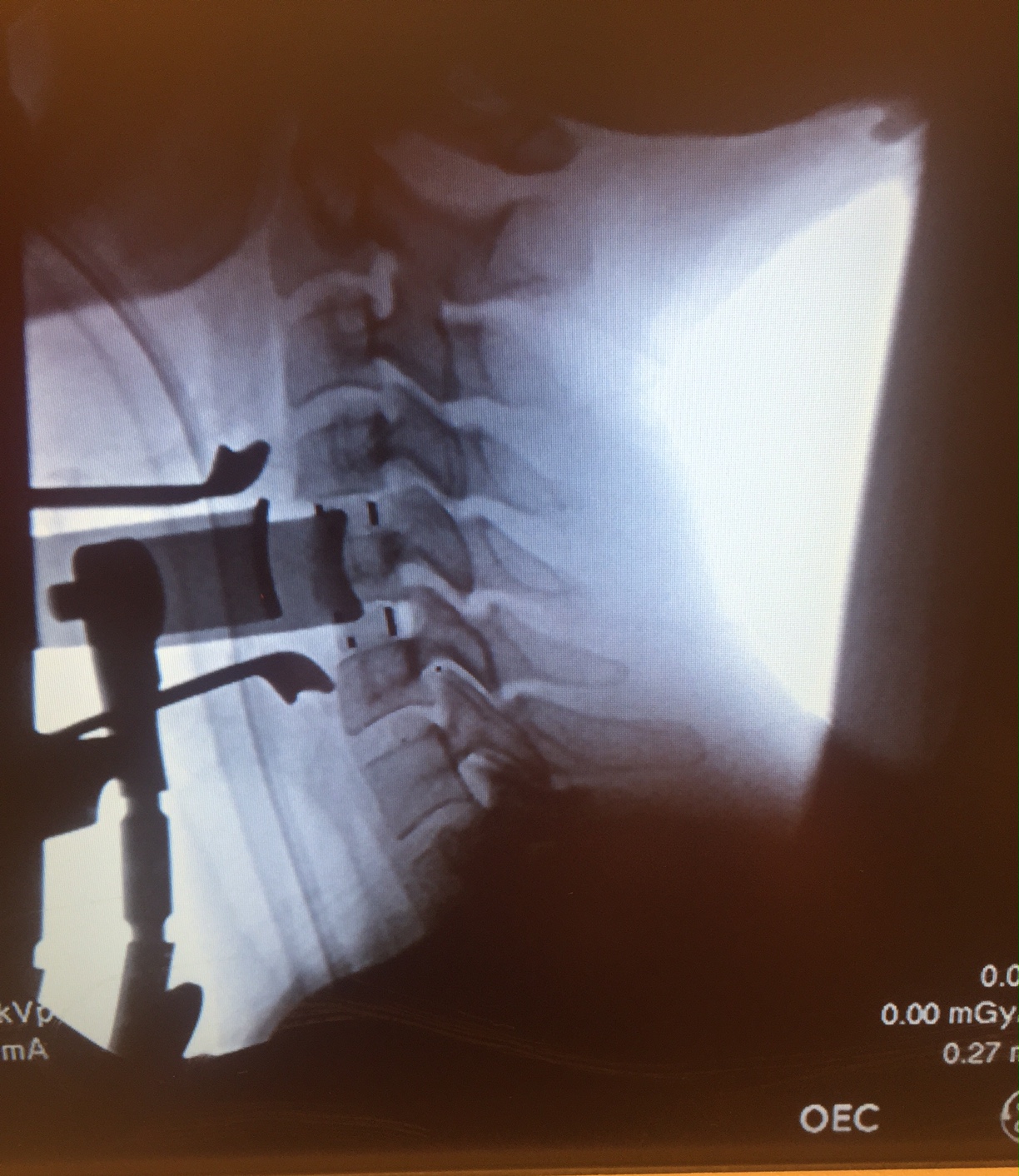
(PRWEB) April 16, 2015 -- On March 26th, 2015, Dr. Mark McFarland became the first surgeon in Virginia to implant Arena-C HA, an innovative spinal implant manufactured with PEEK-OPTIMA® HA Enhanced, a new bioactive material that enhances spinal fusion*. He used this implant in a two-level C4-C5, C5-C6 ACDF LES procedure to treat a patient diagnosed with cervical spinal spondylosis and cervical stenosis. The procedure was completed at Mary Immaculate Hospital in Newport News, VA.
SpineFrontier’s Arena-C HA is an intervertebral spacer made from biocompatible plastic designed to provide mechanical stability for patients who need spine surgery. The implant was designed by SpineFrontier to provide mechanical stability for patients who need spine surgery. This was the first implant ever to be made with Invibio’s PEEK-OPTIMA® HA Enhanced, and was cleared by the FDA in October 2014. Implants are usually made with PEEK-OPTIMA Natural. This product builds on its predecessors. It is manufactured with hydroxyapatite (HA), which is a naturally occurring compound in human bone. The end result is a completely homogenous compound that ensures HA is present on all surfaces of the device.
Dr. McFarland described the case: "Shortening a patient’s recovery time as well as providing a more minimally invasive, LES insertion technique makes this device a valuable addition to our operating room. Once again, SpineFrontier has helped us lead the way in less exposure surgery and improve the lives of our patients."
Dr. McFarland currently practices orthopedic surgery at the Orthopedic and Spine Center in Newport News, VA, where he focuses primarily on the care and treatment of injuries and disorders of the spine. Dr. McFarland graduated from Oklahoma State University Medical School in 1999. He completed his residency in orthopedic surgery at Ohio University and then went on to complete an Orthopedic Spine Surgery Fellowship at the Florida Spine Institute in Clearwater, Florida. Dr. McFarland is a member of the American Academy of Orthopedic Surgeons and the American College of Osteopathic Surgeons. Among other honors, he received the “Graduating Physician Academic Excellence Award.” His research includes studies on vertebral compression fracture outcomes.
More about the Less Exposure Surgery Philosophy: “To tailor treatment to the individual patient to have the quickest recovery and return to a pain-free lifestyle. If surgery is chosen, use Less Exposure Surgery techniques and technologies that lessen exposure proportionate to the problem and the patient, preserve unoffending anatomy, and utilize fundamental principles adapted to new technologies which are safe, easy to adopt, reproducible and lessen blood loss, surgical time, and exposure to radiation. The aim is to achieve maximal effectiveness for a rapid and less painful outpatient recovery.
About SpineFrontier Inc. About SpineFrontier Inc.
SpineFrontier Inc. (http://www.spinefrontier.com) is a growing medical device company that designs, develops and markets implants and instruments utilized for spine surgery. The SpineFrontier team focuses on developing innovative technologies and techniques for spinal surgery based on the Less Exposure Surgery (LES) philosophy. These technologies allow for same day outpatient surgery, due to minimal disruption of normal tissues. SpineFrontier believes LESS is more: LESS time in treatment and recovery is more time in action for our patients and surgeons. SpineFrontier is headquartered in Beverly, MA. It was founded in 2006, and released its first products in 2008. SpineFrontier is a KICVentures portfolio company and an exclusive manufacturer of LES technologies and instruments.
To learn more about SpineFrontier visit http://www.spinefrontier.com.
*Study evaluated the bone on-growth of PEEK-OPTIMA and PEEK-OPTIMA HA Enhanced in a bone defect model in sheep. Study did not use Arena-C or Arena-C HA products Data on file at Invibio. This has not been correlated with human clinical experience.
For more information about PEEK-OPTIMA HA Enhanced, please visit TurbochargeFUSION.com
PEEK-OPTIMA® is a registered trademark of Invibio, Ltd. All rights reserved.
Anders Johnson, SpineFrontier, Inc., http://www.spinefrontier.com, +1 9782323990, [email protected]
Share this article