Atlanta-Based NFANT Labs Receives FDA Clearance for its nfant® Feeding Solution
ATLANTA (PRWEB) October 07, 2015 -- Atlanta-based NFANT Labs LLC today announced that it has received FDA clearance for its nfant® Feeding Solution. Unlike an ordinary baby bottle, nfant® Feeding Solution is the only medical device that can determine an infant’s tongue movement during actual feeding and provide objective, measurable data. Information is then relayed to caregivers through a mobile app and stored in a cloud database. The solution is the first Internet of Things (IoT) medical device focused on babies in the 1,100 Neonatal Intensive Care Units (NICUs) across the United States.
“Since beginning product development just a year and a half ago, we have successfully crossed another hurdle by receiving our FDA clearance,” said Lou Malice, CEO of NFANT Labs. “Up to 70 percent of the 540,000 premature babies born in the U.S. each year experience feeding problems. Until now, physicians and caregivers have not had access to objective feeding data and the power of cloud analytics to enhance decision making.”
Currently, deciding exactly when it is safe to begin oral feeding and determining how to best advance an infant’s feeding is based on a caregiver’s professional experience and trial and error. Now, healthcare teams can use the data from nfant Feeding Solution to help determine when NICU infants are ready to transition from tube feeding to bottle or breast feeding.
"Many infants in the NICU have trouble transitioning from tube feeding to bottle or breast feeding and these problems often lead to delays in patient discharge,” said Dr. Gilson Capilouto, professor in the College of Health Sciences at the University of Kentucky and co-founder of NFANT Labs. “Our product focuses on tongue movement since the tongue plays a major role in safe swallow during feeding. Like any muscle, it’s equipped with strength properties that can be measured and rehabilitated. Through nfant Feeding Solution we have created the first and only medical device that can determine an infant’s tongue movement during feeding and offer a method to capture and return data to the caregiver.”
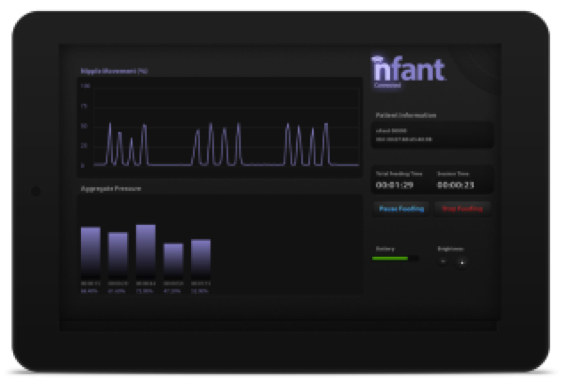
Each nfant Feeding Solution consists of a disposable nfant Coupling that connects the bottle to the nipple. Reusable nfant Sensors then connect to the Coupling and non-invasively measure the baby’s tongue movement while transmitting that data to a smart device for display in real-time. Data is then stored in a cloud database for analysis and retrieval. In addition to real-time analytics, physicians and caregivers can also view an infant’s historical feeding analytics to determine progress.
"There’s no one else in the market doing what NFANT Labs is doing, and I believe that nfant Feeding Solution is going to make a tremendous impact on getting high risk infants to full feeds safely,” said Dr. Peter Giannone, chief, Division of Neonatology at University of Kentucky Children's Hospital, “It provides physicians and caregivers with objective metrics that have never been as readily available. This allows for better decision-making among the entire NICU healthcare team, reduces silos of care and can ultimately improve the outcomes for patients.”
NFANT Labs will start providing NICUs with nfant Feeding Solution this quarter with plans to expand its market in 2016. For more information, visit http://www.nfant.com.
About NFANT Labs LLC:
Founded in 2013, Atlanta-based NFANT Labs is an emerging medical device company focused on improving the outcomes and lives of neonatal infants through internet connected medical devices. Its product, nfant® Feeding Solution, is the first FDA cleared IoT medical device focused on the NICU. Improving the standard of feeding care by collecting objective data and tracking feeding progression has the potential to shorten NICU stays, reduce readmissions and deliver substantial savings. For more information about NFANT Labs, please visit http://www.nfant.com.
Colleen Pinto, AR|PR, http://www.arpr.co, +1 855.300.8209, [email protected]



Share this article