Clinical Study Results of ISS’ Intelligent, Wireless, Left-Heart, Implantable, Hemodynamic Monitor will be Presented at the HFSA 20th Annual Scientific Conference 2016
Michigan, USA (PRWEB) September 15, 2016 -- Integrated Sensing Systems, Inc (ISS) will be presenting the clinical study results of its intelligent, wireless, left-heart, implantable, hemodynamic monitor at the HFSA 20th Annual Scientific Conference 2016. (http://meeting.hfsa.org/)
The poster entitled “An implantable pressure sensor for wireless monitoring of intracardiac pressures_first study in man,” will be presented by Dr. Jacek Baranowski (Linkoping University Hospital, Linkoping, Sweden) on Saturday September 17th at 6:15 pm (Abstract #188).
ISS IHM implant has been designed to monitor filling pressure, heart rate, rhythm and pressure waveforms of the left side of the heart. The main goal of the individualized custom treatment for heart failure patients is to maintain a fine balance between improving cardiac output (blood flow pumped by a ventricle) and sustaining both a reasonable left-heart filling pressure and proper heart rhythm. Thus, monitoring left-heart pressure waveforms is the most desirable parameter for the management of patients with cardiovascular problems.
The Linkoping Hospital and ISS successfully conducted two clinical investigational studies: (1) monitoring left-heart (left atrium or left ventricle) hemodynamics to improve the long-term treatment of congestive heart failure (CHF) patients, and (2) monitoring the left atrium to improve the safety and treatment of LVAD patients.
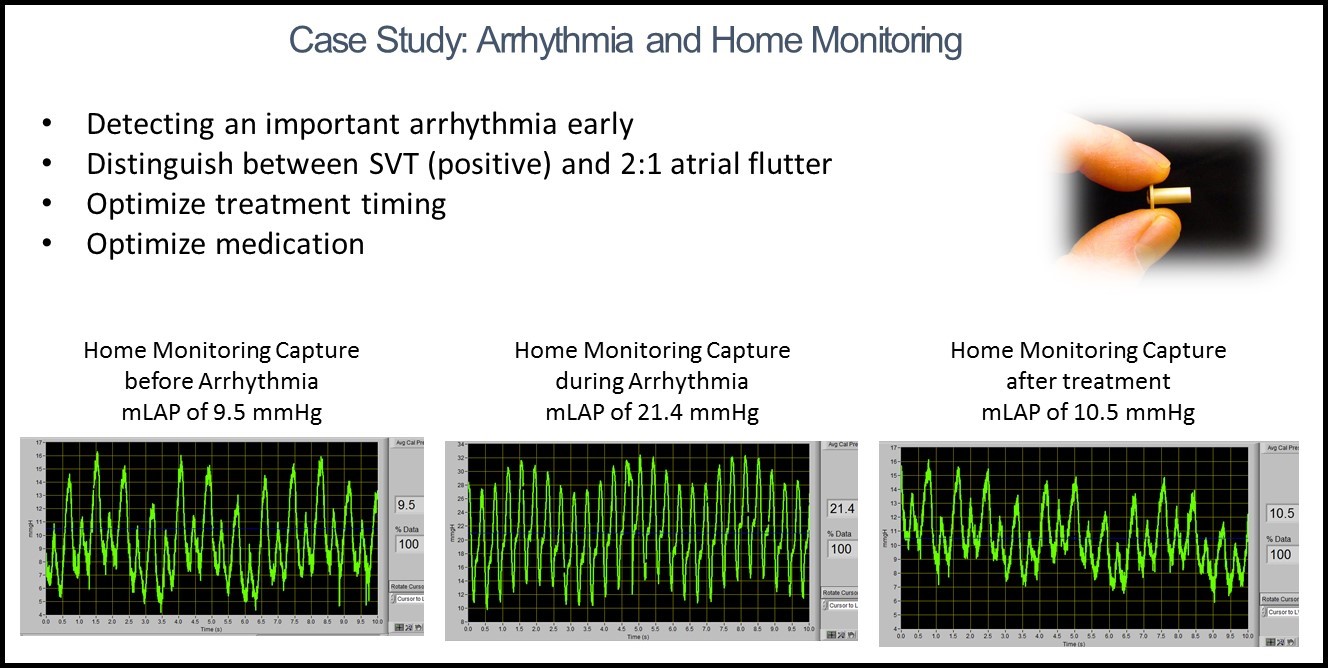
The ongoing CHF clinical study was started in June 2013 and its 6-month follow-up of the last patient was finished in July 2015. During a cardiac surgery, on average only about 8 additional minutes were required to place the ISS implant. The safety results are outstanding and show major improvements in reducing the severe adverse effects. The data further indicates major efficacy trends in the long-term management of cardiac patients. A positive efficacy trend of the ISS’ IHM was improvements in post-op ICU management, including early detection of tamponade and potential for reducing secondary organ failures. After almost 3 years, the results of this study, which met its primary and secondary endpoints, continue to substantiate the high safety profile and potential effectiveness of the ISS IHM as a viable pressure monitoring system for the chronic management of patients with congestive heart failure, arrhythmia, and structural hearts diseases. Compared to the control, patients with the ISS IHM sensor showed shorter duration in hypotension, anuria and oliguria during ICU, and improvement in CHF left ventricle function, symptoms and rate of re-hospitalization. Due to instantaneous filling pressure monitoring, patients with the ISS IHM sensor further benefited from rapid effective tailoring and adjustment of medications, early detections of different types of arrhythmias, in particular fibrillation episodes, detection and management of mitral regurgitation, better management of hypertension, and early warning of dangerous onset of conditions such as tamponade.
ISS has developed two categories of products: IHM sensors that are implanted adjunct to a planned thoracic surgery and implants that are delivered percutaneously via catheter-based techniques in either the pulmonary artery (PA) or left atrium during a stand-alone procedure. ISS IHM is composed of three components: a Telemetric Implant, a Monitoring Unit, and the Database Management system for internet-based worldwide access.
ISS IHM product is restricted under United States Federal Law to investigational use only, and is being filed for CE Mark approval in Europe.
About Integrated Sensing Systems, Inc.:
ISS is a leader in advanced MEMS technologies for design and manufacturing of medical devices. Founded in 1995, ISS is one of the oldest independent medical MEMS companies in the United States. ISS operates a comprehensive, state-of-the-art MEMS fabrication facility located near Ann Arbor, Michigan. ISS is currently certified for ISO 9001:2008, EN13980:2002 for ATEX (intrinsically safe products), and ISO13485:2003 for Class III medical devices. ISS is a vertically integrated company, dedicated to developing and manufacturing system-level products based on MEMS technology (MEMS Inside), please visit: http://mems-iss.com/titan-wihm/
Source: Integrated Sensing Systems, Inc. (ISS)
Contact:
Nader Najafi, Ph.D.
Integrated Sensing Systems Inc. (ISS)
391 Airport Industrial Dr., Ypsilanti, MI 48198
Tel: (734) 547-9896 Ext. 103
Fax: (734) 547-9964
Email: nader(at)mems-iss(dot)com
Nader Najafi, Integrated Sensing Systems, Inc., http://www.mems-iss.com, +1 734-547-9896 Ext: 103, [email protected]
Share this article