FDA Approves New Infusion Catheter
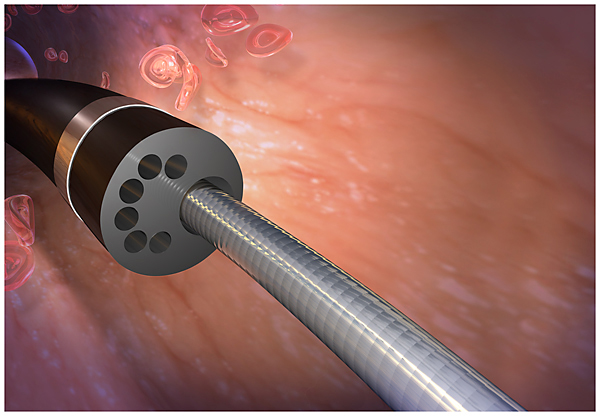
Gilbert, AZ (PRWEB) April 29, 2014 -- The Translational Research Institute “TRI” is pleased to announce that the Food and Drug Administration recently approved TRI’s new ND™ Infusion Catheter. On November 15th 2013, the FDA cleared the use of the patented ND™ Infusion Catheter for “infusion of physician-specified fluids.” The ND™ Infusion Catheter is designed to isolate a specific vascular treatment region from blood flow during infusion while delivering the physician specified fluid.
TRI’s Chief Executive Officer Nabil Dib MD, MSc stated, “This breakthrough technology has two significant scientific advancements. The multi-lumen channels provide the smallest luminal diameter available in catheter technology today. The micro channels offer better mixing and dispersion of therapeutic agents, which potentially enhances efficacy.
Additionally, the catheter’s balloon is uniquely designed to minimize pressure on the vessel wall during inflation, conceivably decreasing incidence of vascular trauma compared to conventional technology. Since the balloon adapts to different vessel diameters, inventory is reduced, thus decreasing costs” Dr. Dib continued, “This innovative technology provides a strategic tool for physicians to deliver better patient care. We are delighted to have gained FDA approval so quickly. TRI’s ability to advance from concept to approval in 31 months is a direct result of our Translational Research infrastructure which is elemental to our ability to expedite scientific discovery for patients at a decreased total cost.”
ABOUT TRI
TRI is a privately held, medical device development company. TRI is dedicated to providing a pathway to regulatory approval that is efficient, predictable and cost effective.
Media inquiries regarding TRI, its capabilities and for additional information regarding the ND™ Infusion Catheter contact:
DeAnn Dana
Email: DDana(at)TRImedical(dot)com
Phone: 480.309.2884
http://www.trimedical.com
Gregory Patterson, Andale Advertising, http://trimedical.com, +1 (602) 501-0772, [email protected]
Share this article