Mindray Introduces Specialized Transducer and Enhanced iNeedle on TE7 Touch Enabled Ultrasound System for Anesthesia Applications
Mahwah, NJ (PRWEB) October 22, 2016 -- In conjunction with the American Society of Anesthesiologists (ASA) meeting in Chicago opening today, Mindray announces the introduction of its new L20-5s ultra high frequency linear transducer with enhanced iNeedle™ visualization technology. The TE7 Touch Enabled Ultrasound System, designed specifically for the demanding point-of-care (POC) imaging environment, offers intuitive tablet-like operation, superior image quality with one-touch image optimization, and exam presets to improve both diagnostic confidence and efficiency.
“The L20-5s is considered to be a breakthrough transducer in the ultrasound industry,” said Glen McLaughlin, Ph.D., chief product officer for Mindray. “Coupling this ultra-high frequency transducer with our iNeedle technology is particularly useful in anesthesia POC to provide increased visualization in the area of interest and enhanced needle detection. This can assist in reducing stress on the patient and help reduce procedure time.”
Mindray continues to innovate ultrasound technologies to meet the demands of the fast-paced anesthesia environments. Unlike many POC systems, the TE7 cuts through the clutter with no keyboard, streamlining clinical workflow with intuitive gestures. The TE7 already goes further with superior image quality, up to three active transducer connectors, slim profile ergonomic cart and a rich set of applications including cardiac functionality with continuous wave (CW) Doppler and a transesophageal echocardiography (TEE) transducer. The new ultra high frequency transducer and improvements in the iNeedle application demonstrate Mindray’s continuing focus on responding to the needs of the anesthesia professional, from needle placement to cardiac monitoring in the cardiovascular OR.
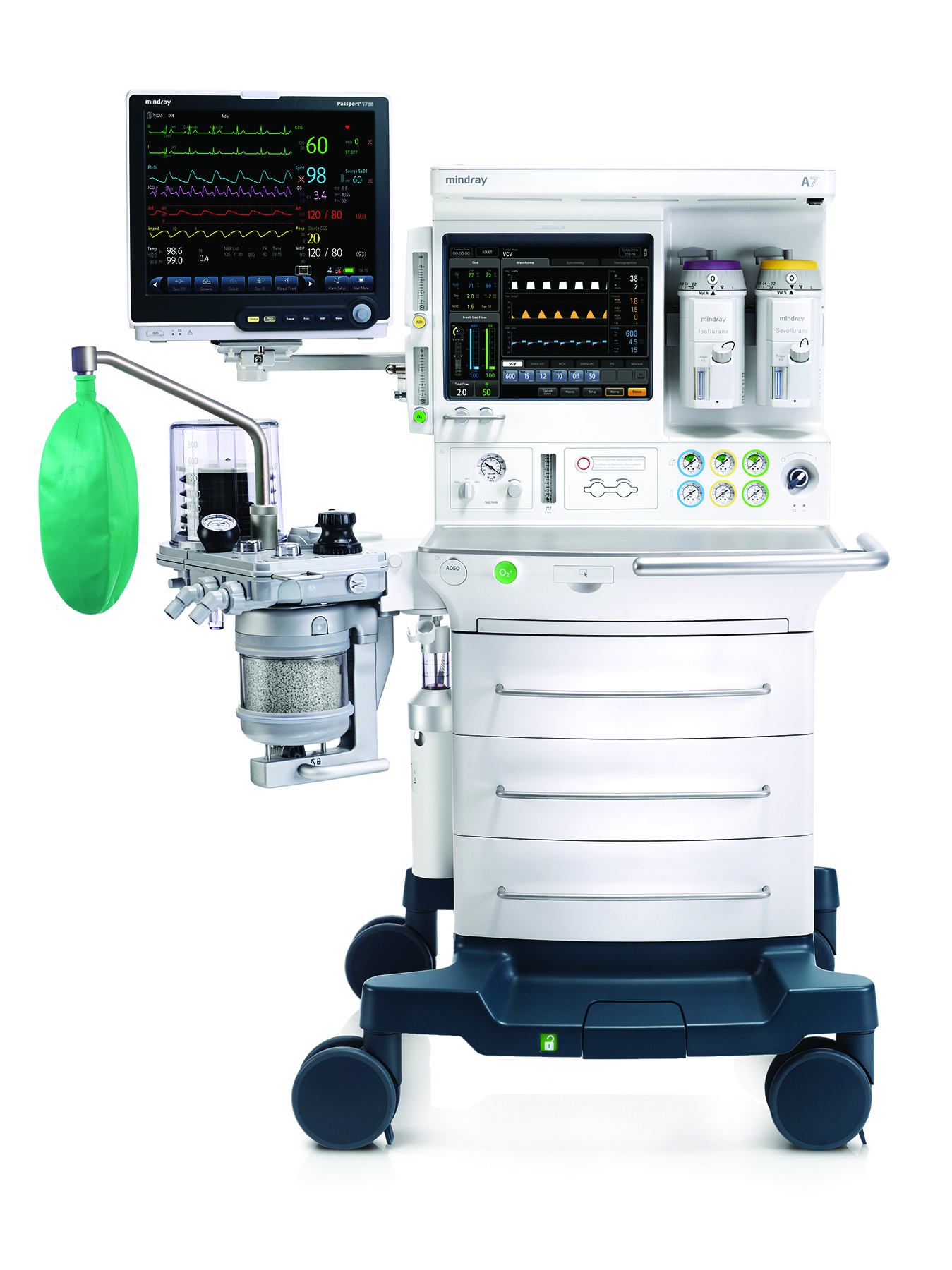
At ASA, the TE7 will be shown with in-booth scanning. Also at this important annual meeting, Mindray will be showcasing the A7 Anesthesia Workstation with the Optimizer™ suite to assist in the reduction of anesthetic gas use and the new Passport Series monitors with the innovative T1 Transport Monitor/Module.
“Mindray has become a leader in the complete perioperative continuum with point-of-care ultrasound, anesthesia delivery, and comprehensive monitoring solutions,” said Wayne Quinn, president of Mindray North America. “According to third-party U.S. market reports, Mindray continues to experience significant growth with its well-received A-Series anesthesia machines. The new capabilities for our popular TE7 ultrasound system further reinforce our commitment to the broader anesthesia market. The OR allows us to showcase all of our solutions, coming together to meet the needs of the anesthesia provider.”
About Mindray
Mindray is a leading developer, manufacturer and marketer of medical devices worldwide. Mindray maintains its global headquarters in Shenzhen, China; North American headquarters in Mahwah, New Jersey; and multiple development facilities and offices in major international markets including an ultrasound Innovation Center in Silicon Valley. Mindray supplies a broad range of products across three primary business segments, patient monitoring and life support, in-vitro diagnostics, and medical imaging systems. For more information, please visit http://www.mindray.com.
Peter Collins, PC Media Relations, +1 (908) 499-1200, [email protected]
Share this article