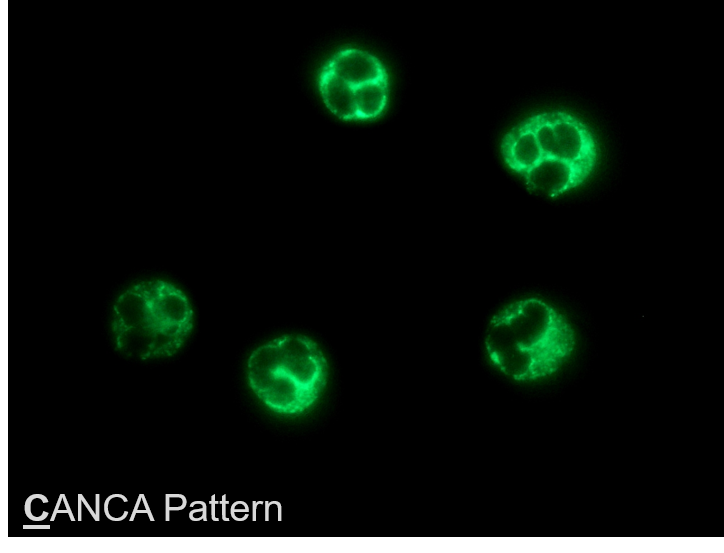
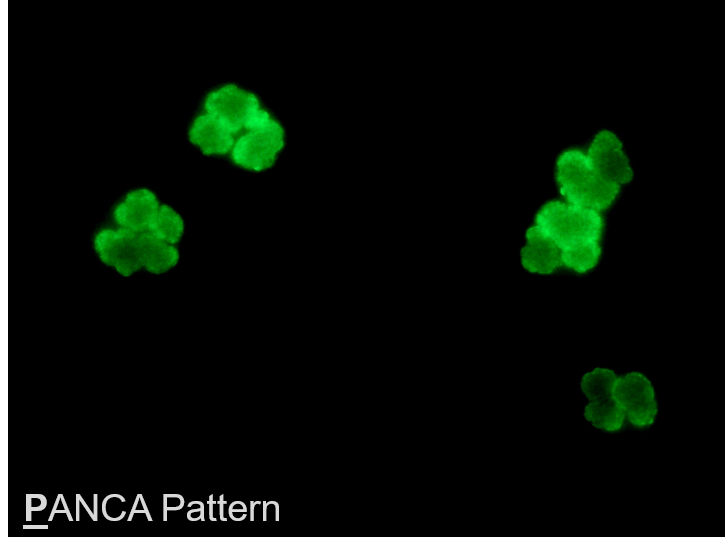
FDA Clears Automated Vasculitis Test from AESKU
WENDELSHEIM, Germany and OAKLAND, Calif. (PRWEB) April 04, 2018 -- AESKU.GROUP – a science-focused manufacturer of autoimmune, allergy, and infectious disease diagnostics – received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for AESKUSLIDES ANCA Ethanol, and ANCA Formalin with the new HELIOS AUTOMATED IFA SYSTEM, and will be followed by esophageal and tissue section slides.
"These are 3rd and 4th FDA cleared assays on the HELIOS, after ANA HEp-2 [in 2016] and nDNA [cleared last month], thanks to our great Regulatory Affairs team at AESKU. With four kits, the HELIOS has the largest FDA cleared IFA menu of any platform and is the only sample-in/result-out system, with half the footprint. Through integrated design and pattern recognition software, the HELIOS system solves the subjectivity, labor, and traceability problems of IFA so laboratories can use the highest quality tests and ultimately improve patient outcomes," said Dr. Torsten Matthias, founder and CEO of the AESKU.GROUP.
"ANCA-associated vasculitis assays are one of the rare STAT autoimmune tests. When a hospital needs to distinguish between AAV and infection, automated IFA will save precious minutes in a very critical situation and help physicians make the appropriate therapeutic decision. The HELIOS, combined with our large IFA portfolio and AESKU.LAB automation management tool can easily fit into a distributed healthcare system bringing testing closer to patients, and improving access. We look forward to making the kit and system available to US healthcare providers with the help of Grifols, our exclusive distribution partner in the US,” said Bruno Larida, Vice-President AESKU.INC.
The HELIOS is a revolutionary platform that both processes and analyzes patient samples in a single run. It is the first and only clinical platform to offer this level of IFA automation, overcoming the previous limitations of IFA testing and letting users return to the gold-standard method for Autoimmunity screening. Over 300 HELIOS systems are in clinical use worldwide, in over 40 countries including the USA.
The HELIOS system and associated FDA cleared kits will be displayed at the American Association of Clinical Chemistry meeting.
For additional information please visit http://www.whatishelios.com
About AESKU. Diagnostics GmbH & Co. KG
AESKU.GROUP (http://www.aesku.com) is headquartered in Wendelsheim, Germany and is made up of the following divisions: AESKU.DIAGNOSTICS, AESKU.THERAPY (Autoimmune Therapies), AESKU.SYSTEMS (Lab Automation), AESKU.INC (North American headquarters in Oakland, CA), AESKU.NY (IFA Manufacturing and Research in Buffalo, NY).
Besides its own outstanding commitment to research and development, AESKU.GROUP initiated and supports the AESKU.KIPP INSTITUTE, a non-profit organization, primarily active in basic research and interdisciplinary knowledge transfer in autoimmunity.
Benjamin Lack, Aesku Diagnostics, http://www.aesku.com, +1 +1-510-761-9162, [email protected]
Share this article