Beneufit Inc., a Digital Health company, announced today that the United States Patent and Trademark Office (USPTO) has issued a patent for its smartphone-based symptom measurement technology.
SAN FRANCISCO, April 21, 2022 /PRNewswire-PRWeb/ -- The USPTO has issued U.S. Patent No. US11,266,341 entitled "Measuring Dynamic Body Movement" for their innovative Tracer™ technology that measures and scores movement disorder symptoms objectively with smartphone video and machine learning techniques. The aim of the technology is to empower patients and caregivers to measure disease state objectively from the comfort of their own home.
Currently the gold standard of measuring disease state in movement disorders is through assessment surveys administered by clinicians who rate the patient's presentation of motor symptoms. These surveys have limited sensitivity because judging motor performance is often subjective and patient scores can vary depending on the clinician administering them. "The reliability of clinical assessments vary considerably with inter-rater variability, which can adversely affect the outcomes in clinical trials. Using technology which can measure even the smallest clinical benefits not noticeable by clinical raters can make the difference in a medication or medical device getting FDA approval. By using Tracer™ we mitigate the risk of bias, variability and human error in clinical trials and evaluating the benefit from medical treatment," says movement disorder specialist and patent co-author Jerome Lisk, MD.
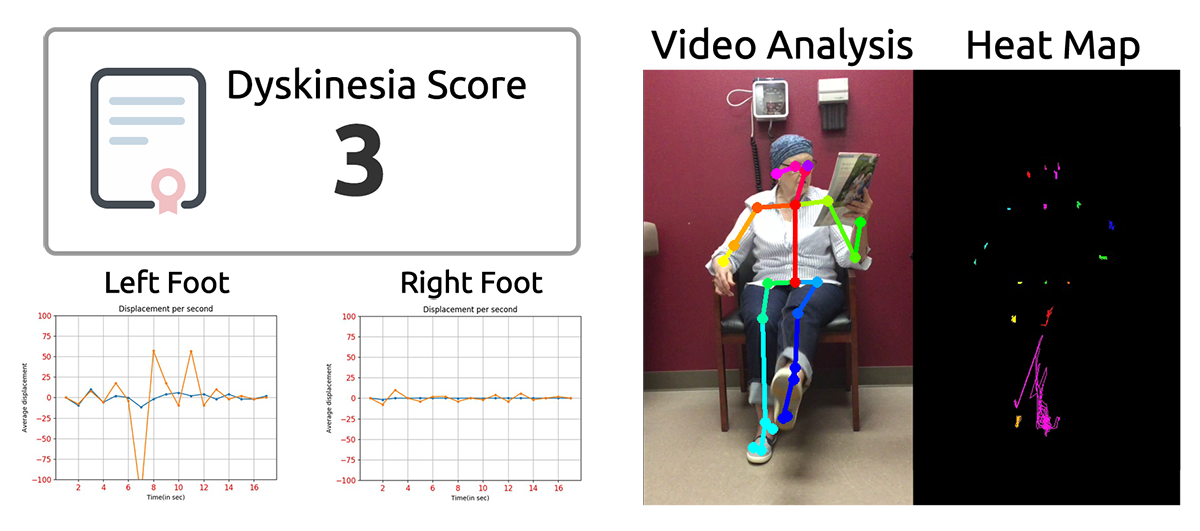
The original proof of concept was built to measure and score Parkinson's disease dyskinesia, a condition that is a side effect of medication and potentially involves all parts of the body. The advantages Tracer™ has over existing wearable sensor systems is that it does not require any devices to be worn by the patient, and it is able to detect motor movement across the patient's entire body. Existing technologies require a sensor to be placed on the part of the body being measured. Motor symptoms such as dyskinesia involve involuntary gross movements that are hard to detect with individual sensors without the context of how other parts of the body are moving that Tracer™ captures by design.
"We're extremely proud of this innovation and excited about how it will help so many in medical research and industry," explained CEO Jeff Broderick. "And we're grateful for the people at the U.S. Food & Drug Administration (FDA) who have encouraged us to deliver Tracer™ clinical outcome assessments (COA) to the drug development ecosystem. Our vision aligns perfectly with the FDA's 21st Century Cures Act which seeks to modernize clinical trial design and include the use of patient-generated COAs."
The technology is currently utilized by two organizations to improve treatment plans for their respective patient populations. One organization, who remains confidential, seeks to reduce infant suffering. An app enables parents to get an instant analysis of their baby's movement, with accompanying data which can be shared with their pediatrician.
In another project, Tracer™ is being used by the TESS Research Foundation to find a biomarker for SLC13A5 Epilepsy. In addition, Tracer™ will generate digital assets that will be used to quantify assessment of body movement. "One symptom of SLC13A5 Epilepsy is a movement disorder that significantly affects the health and quality of life of our affected families. We are hopeful that Tracer will help accelerate drug discovery for our SLC13A5 community by providing an objective and reliable metric for movement disorder biomarkers," says Kimberly Nye, Founder and Executive Director of the foundation.
About Beneufit, Inc.
Beneufit provides innovative digital health solutions to organizations using patient-generated data to advance research, improve outcomes and increase efficiencies.
Contact:
Jeff Broderick
[email protected]
About TESS Research Foundation
TESS Research Foundation is driving research to cure SLC13A5 Epilepsy. To achieve our goals, TESS funds cutting-edge research, provides support to families affected by SLC13A5 Epilepsy, and increases awareness about this severe neurological disorder.
Contact:
Lindsay Okamoto
[email protected]
Media Contact
Jeff Broderick, Beneufit, Inc., 1 (415) 741-1660, [email protected]
SOURCE Beneufit, Inc.

Share this article