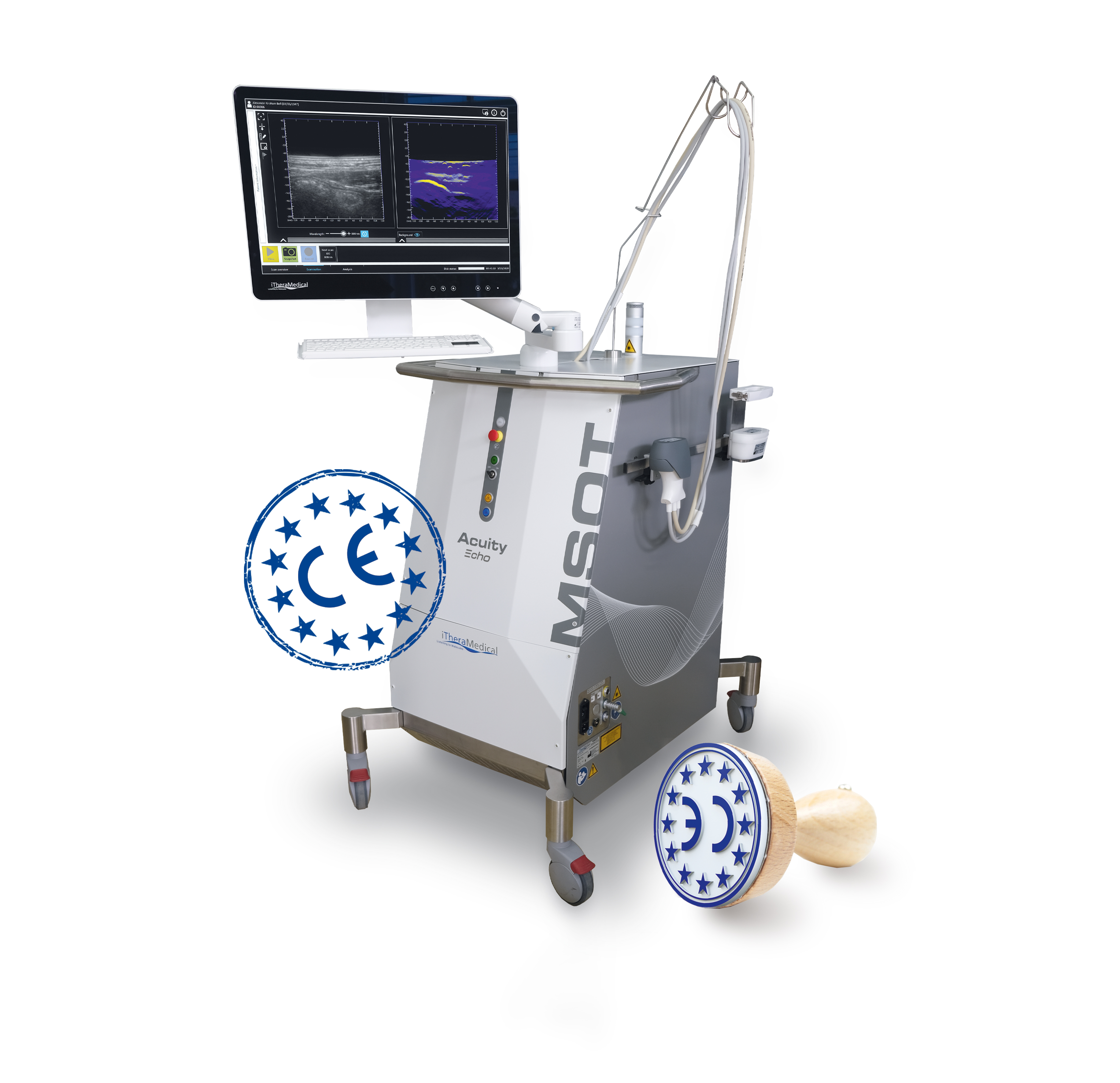
CE mark awarded to iThera Medical's MSOT Acuity Echo imaging system - a major milestone for Horizon 2020 Project EUPHORIA
A CE Mark has been awarded to iThera Medical's MSOT Acuity Echo imaging system, indicating conformity with health, safety and environmental protection for medical devices sold in Europe. The MSOT Acuity Echo combines optoacoustic and ultrasound tomography. The combined technology was optimized within the EU Horizon 2020-funded project EUPHORIA (http://www.euphoria2020.eu), where the new device is also being tested as a clinical tool to non-invasively monitor inflammatory bowel disease.
MUNICH, Germany, June 9, 2021 /PRNewswire-PRWeb/ -- "We are very excited about the CE certification of our MSOT Acuity Echo system. The integration of ultrasound imaging is key for clinical users of optoacoustics" said Christian Wiest, CEO and co-founder of iThera Medical. Dr Philipp Bell, Chief Commercial Officer at iThera and Coordinator of EUPHORIA said "Obtaining the CE mark is a major milestone for the EUPHORIA project. We see huge potential for the MSOT Acuity Echo and the opportunity for non-invasive patient imaging that it presents".
MSOT (multispectral optoacoustic tomography) is based on the photoacoustic effect (the emission of acoustic signals following light absorption). MSOT has the unique capability to visualize optical contrast at high resolution, in deep tissue in real time. By tuning the wavelength of the excitation laser, it can discriminate tissue chromophores that have a characteristic spectrum in the near-infrared; particularly hemoglobin, lipids, collagen, and water. The combination of optoacoustic imaging with ultrasound allows the clinician to identify relevant anatomical structures based on the ultrasound image and then probe the tissue of interest for functional and molecular biomarkers using MSOT.
The technology behind iThera's MSOT Acuity Echo was optimized as part of the EU Horizon 2020 EUPHORIA Project. The project aims to test the efficacy of the new system as a clinical tool for non-invasive monitoring of Inflammatory bowel diseases (IBD), including Crohn's disease and ulcerative colitis. A multi-site clinical study in people with IBD is currently underway in four hospitals in Germany and Italy.
Initial research suggests there are a wide range of potential clinical applications for MSOT and the MSOT Acuity Echo. With the receipt of the CE mark, clinical researchers can now readily explore the clinical value of MSOT, even following the imminent introduction of the new Medical Device Regulation.
About the EUPHORIA Project
The EUPHORIA Project is an EU funded Fast Track to Innovation project led by iThera Medical. The Project also involves University Hospital Erlangen (Germany), IMASONIC SAS (France), Rayfos Ltd (UK) and Pintail Ltd (Ireland). The Project is focused on optimizing iThera Medical's MSOT Acuity Echo, testing its efficacy in a large clinical study, achieving CE marking for the device, validating clinical utility and demonstrating benefits for those with IBD. The consortium is also exploring potential health economic impacts and plans are being developed to bring this novel technology to the market-place and to the clinic. You can find out more about the EUPHORIA Project at https://euphoria2020.eu/.
For further information, please contact:
Nora Ward
Email: [email protected]
The EUPHORIA Project has received funding from the European Union's Horizon 2020 research and innovation programme under grant agreement No 830965. The material presented and views expressed here are the responsibility of the author(s) only. The EU Commission takes no responsibility for any use made of the information set out.
SOURCE iThera Medical

Share this article