Clinerion and iClusion partner to boost the identification and recruitment of more patients into clinical trials
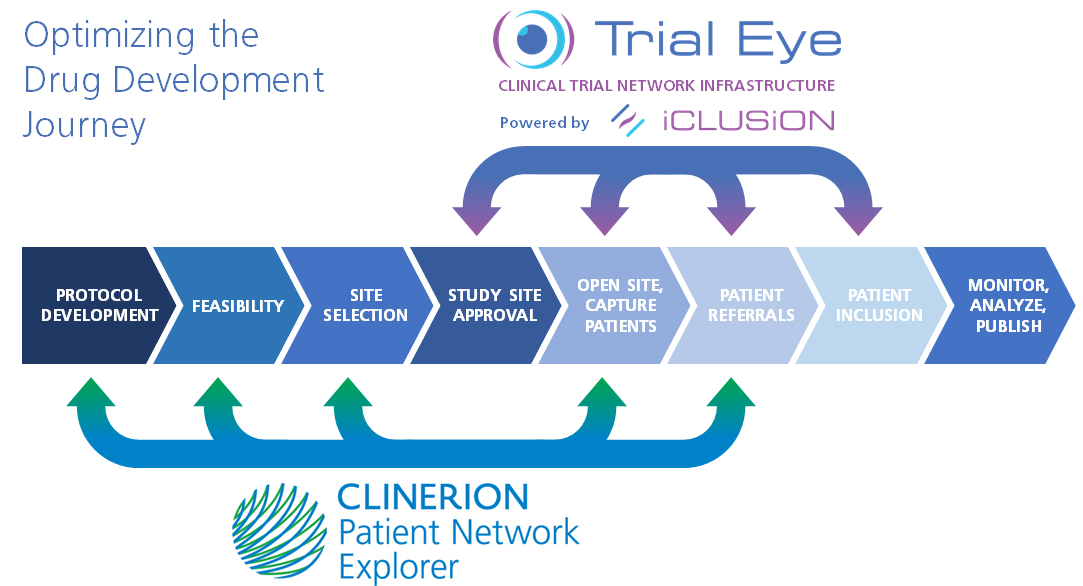
Clinerion's Patient Network Explorer and iClusion's Trial Eye platforms both connect clinical trial sponsors with networks of partner hospitals, giving trial managers the ability to include eligible patients for trials more quickly and efficiently than before. Treating physicians and patients benefit from increased treatment options and quicker access to new medical interventions.
BASEL, Switzerland, March 3, 2020 /PRNewswire-PRWeb/ -- Together, the partners' joint mission is to increase patient participation in studies to its full potential by optimizing protocol design, enabling study sites to efficiently start up their trials locally and plan and meet realistic patient recruitment and therefore total timeline targets.
This new clinical trial optimization partnership also facilitates a total study site network activation for all trials, beyond the study sites already chosen to participate, by facilitating online referral and activation services.
iClusion makes clinical research accessible for physicians and their patients by connecting sponsors and their research protocols with research sites. Connecting all stakeholders and automating standardized local approval procedures in one networked platform accelerates total study timelines and makes innovative products available to more patients faster.
Clinerion makes real-world patient data accessible for analysis so that study protocols can be optimized with real patients in mind and real-world patient data can drive study feasibility and patient recruitment.
Both iClusion and Clinerion maintain cloud platforms which bring together participants from across the clinical study ecosystem, comprising sponsors, hospitals and clinicians, and treating physicians and patients. However, the partners offer complementary support and solutions throughout the product development process.
"Never in history has the R&D engine in pharmaceuticals been more productive, driving a pool of promising new agents," says Edwin Klumper, CEO of iClusion. "The growing pipeline of targeted and personalized medicines puts tremendous pressure on more trials to be conducted in smaller, more well-defined patient populations. As a result, patient recruitment has become the bottleneck, nowadays, which is delaying progress and withholding many patients from the benefits of participation. I am pleased that Clinerion and iClusion are partnering to start tackling the identification and recruitment of the right patients into the right studies to bolster trial conduct and innovation."
"Our collaboration re-emphasizes that patients are at the heart of clinical development and clinical trial conduct," says Ian Rentsch, CEO of Clinerion. "Our combined services cover trials operations from protocol design through operational feasibility, site selection, local approvals, startup of study sites and patient recruitment. This partnership is therefore ideally positioned to optimize and advance protocols faster, reach more study sites and offer more patients the possibility to participate in research."
Clinerion and iClusion will present their combined offerings at the upcoming SCRS's European Site Solution Summit in Lisbon, Portugal, on March 9-10, 2020. Clinerion will be at Booth #25.
European Site Solution Summit website: http://www.eusitesolutionssummit.com.
About Clinerion
Clinerion accelerates clinical research and medical access to treatments for patients. We use proprietary technologies for analysis of patient data from our global network of partner hospitals. Clinerion's Patient Network Explorer radically improves the efficiency and effectiveness of clinical trial recruitment by offering data-driven protocol optimization, site feasibility evaluation and real-time patient search and identification to match patients to treatments. Our technology solution provides real-world evidence analytics for medical access. Clinerion facilitates the participation of partner hospitals in leading-edge, industry-sponsored trials and time savings in patient recruitment. We create innovative and disruptive fit-for-purpose solutions which enable pharmaceutical companies to shorten patient recruitment and save costs by streamlining operations and leveraging strategic intelligence. Clinerion's proprietary Big Data analytics technologies leverage real-time data from electronic health records which comply with international patient privacy and data security regulations. Clinerion is a global data technology service company headquartered in Switzerland.
Clinerion website: http://www.clinerion.com
Clinerion's Patient Network Explorer: http://www.clinerion.com/index/OverviewOurSolutions/ClinerionPatientNetworkExplorer
For more information, please contact:
Le Vin Chin
Director, Head of Marketing & Communications
Clinerion Ltd
Elisabethenanlage 11, 4051 Basel, Switzerland
Tel.: +41 61 865 60 54
[email protected]
About iClusion
Clinical trials don't just advance science; they also serve as in-the-moment treatment alternatives for patients. iClusion aims to make the clinical trial process faster, more effective, and more accessible for cancer patients and oncologists. The Trial Eye platform of iClusion connects study sponsors, hospitals, physicians, and their patients in a closed-loop ecosystem, non-existing today. Rather than each group remaining siloed, iClusion's clinical study infrastucture invites various stakeholders to connect and interact speeding up clinical trial processes and patient recruitment and therefore the entire drug development process. iClusion believes its new model is nothing short of revolutionary. It's the only initiative designed from the viewpoint of the 2 most important stakeholders within clinical trials; the patient and their treating physician. If they have easy access to clinical trials and can participate, drug development can speed up. The Social Impact and Goal is that each cancer patient should be offered the opportunity to participate in an experimental treatment whereas today only 5% of patients do get this opportunity to a last life line.
iClusion website: http://www.iclusion.com
For more information, please contact:
Hanneke Janssen
iClusion
General Manager
iClusion BV
Groot Handelsgebouw, Stationsplein 45 (Unit A4.004), 3013 AK, Rotterdam, The Netherlands
Tel.: 0031 6 20684080
[email protected]
SOURCE Clinerion Ltd.






Share this article