Experience with BioTrace Medical's Tempo Temporary Pacing Lead in Management of Bradyarrhythmia Presented at the American Heart Association's Scientific Sessions 2021
Single-center retrospective analysis further demonstrates the Tempo Lead is safe and effective for temporary pacing and ambulation in cardiac electrophysiology applications
MENLO PARK, Calif., Nov. 15, 2021 /PRNewswire-PRWeb/ -- BioTrace Medical, Inc., the leader in innovative temporary cardiac pacing technology, today announced the company's Tempo® Temporary Pacing Lead was featured in a clinical study presented at the American Heart Association's Scientific Sessions 2021, held November 13-15 in Orlando, Florida.
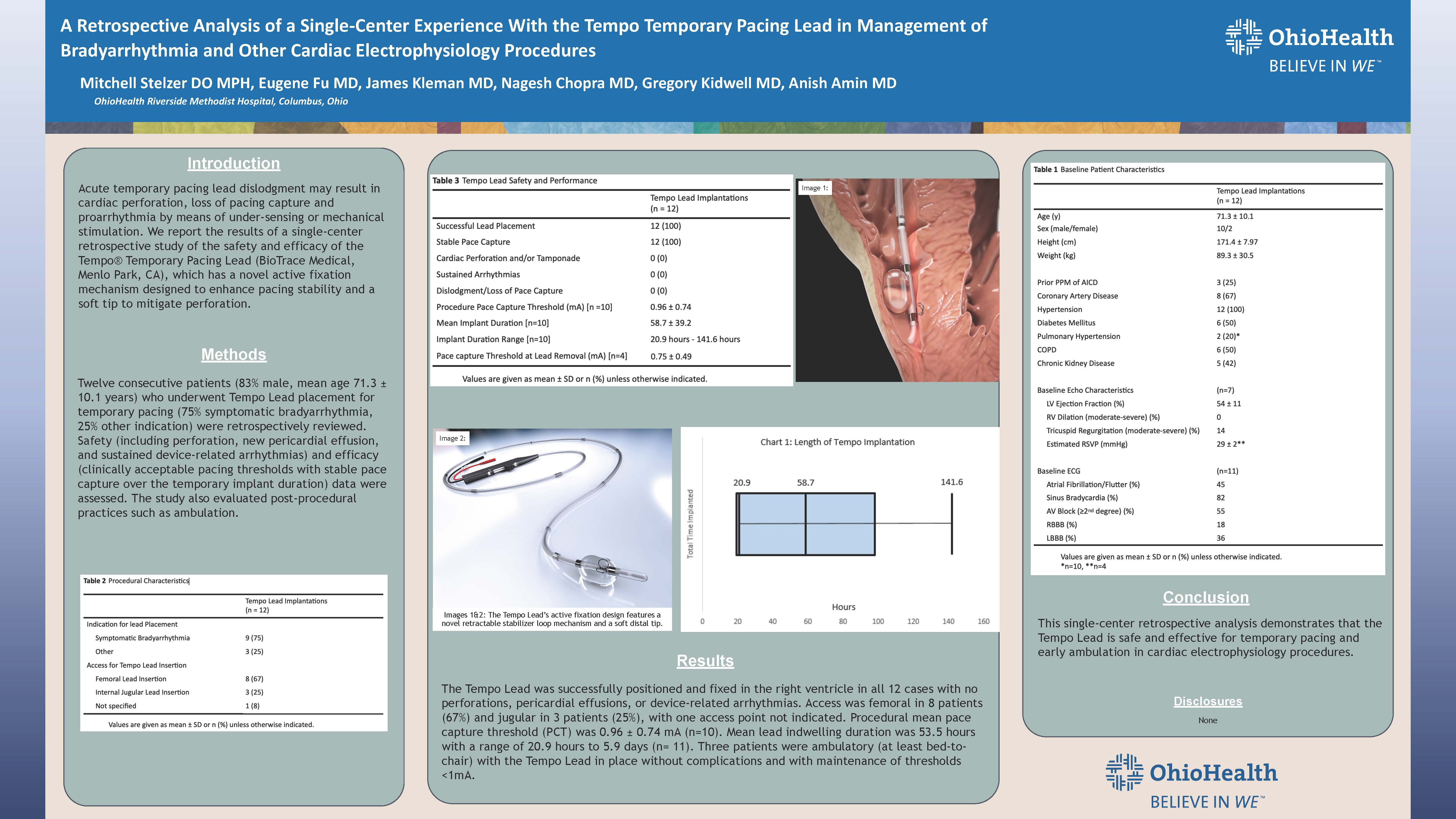
The study, titled "A Retrospective Analysis of a Single-Center Experience with the Tempo Temporary Pacing Lead in Management of Bradyarrhythmia and Other Cardiac Electrophysiology Procedures", analyzed a series of twelve consecutive patients who underwent Tempo Lead placement for temporary pacing at OhioHealth Riverside Methodist Hospital in Columbus, Ohio.
Twelve consecutive patients (83% male, mean age 71.3 ± 10.1 years) who underwent Tempo Lead placement for temporary pacing were retrospectively reviewed (75% symptomatic bradyarrhythmia, 25% other indications including permanent lead malfunction, permanent pacemaker infection, and complete heart block.) The Tempo Lead was successfully positioned and fixed in the right ventricle in all twelve cases with no perforations, pericardial effusions, or device-related arrhythmias. Access was femoral in eight patients (67%) and jugular in three patients (25%), with one access point not indicated.
Procedural mean pace capture threshold (PCT) was 0.96 ± 0.74 mA (n=10). Mean lead indwelling duration was 53.5 hours with a range of 20.9 hours to 5.9 days (n= 11). Three patients were ambulatory (at least bed-to-chair) with the Tempo Lead in place without complications and with maintenance of thresholds <1mA.
The Tempo Lead's novel design – featuring retractable stabilizer loops and a soft tip – minimizes the risks of dislodgment and perforation, ensures stability during rapid pacing, and facilitates patient ambulation in recovery.
"The Tempo Lead is very useful for the support of temporary bradyarrhythmias that require reliable early- or medium-term temporary pacing as a bridge to resolution or permanent pacemaker implantation," said Anish K. Amin, MD, cardiac electrophysiologist at OhioHealth Riverside Methodist Hospital. "Its unique design provides temporary pacing without the risk of dislodgment, loss of pace capture, or perforation inherent in conventional temporary leads."
With U.S. FDA clearance and CE certification, the Tempo Lead is the only active fixation temporary pacing lead available in the world. The Tempo Lead has been used in more than 12,000 U.S. procedures with an excellent safety and performance profile, as demonstrated by reported "real-world" 0.04% cardiac perforation rate and a 0.11% intermittent/loss of pace capture rate.1 (Conventional temporary pacing leads exhibit cardiac perforation rates ranging from 0.6% to 4.0%, and dislodgment/loss of pace capture rates between 10% and 37%.2)
About BioTrace Medical
BioTrace Medical, Inc., a private venture-backed company based in Menlo Park, California, is dedicated to reinventing temporary pacing to improve patient outcomes and reduce hospital costs. For more information, visit http://www.biotracemedical.com.
(1) Company data on file as of October 2021.
(2) Metkus, et al. Chest. 2019; Lopez, et al. Rev Esp Cardiol. 2004; Betts, Postgrad Med J. 2003; Jowett, et al. Postgrad Med J. 1989; Austin, et al. Am J Cardiol. 1982; Gammage, Heart 2000; Lumia et al. Chest 1973
APM042-01.A
Media Contact
Kevin Tausend, BioTrace Medical Inc, +1 6507795124, [email protected]
SOURCE BioTrace Medical Inc

Share this article