InfraScan Inc. Announces Issuance of US Patents for Brain Diagnosis
Latest patents expand near infrared platform capabilities related to trauma and stroke diagnosis
PHILADELPHIA, Aug. 3, 2020 /PRNewswire-PRWeb/ -- InfraScan, a medical device firm specializing in brain injury and stroke diagnostic products, announced today the issuance by the US Patent Office of three additional patents that cover internal bleeding, brain trauma and stroke detection technologies.
The first patent for invention number US 2020/0229712 A1 is for an optical probe for oximetry measurements. The second patent for invention number US 2020/0170554 A1 is for a non-invasive device for brain edema and autoregulation monitoring device and method. The patent was developed in collaboration with Drexel University, which owns the patent and licensed it exclusively to InfraScan. The third patent for invention number US 10,292,598 B2 is for devices and methods for detection of internal hematoma. This patent was developed in Hadassah Hospital in Israel, which licensed it exclusively to InfraScan.
"The latest patents make claims in connection with near-infrared technology that allows local hypoxia and edema detection in addition to hemorrhage detection," said Baruch Ben Dor, PhD, president and CEO of InfraScan. "Detecting changes from normal baseline as well as monitoring those parameters in trauma and stroke victims is crucial. It further expands our technology platform portfolio."
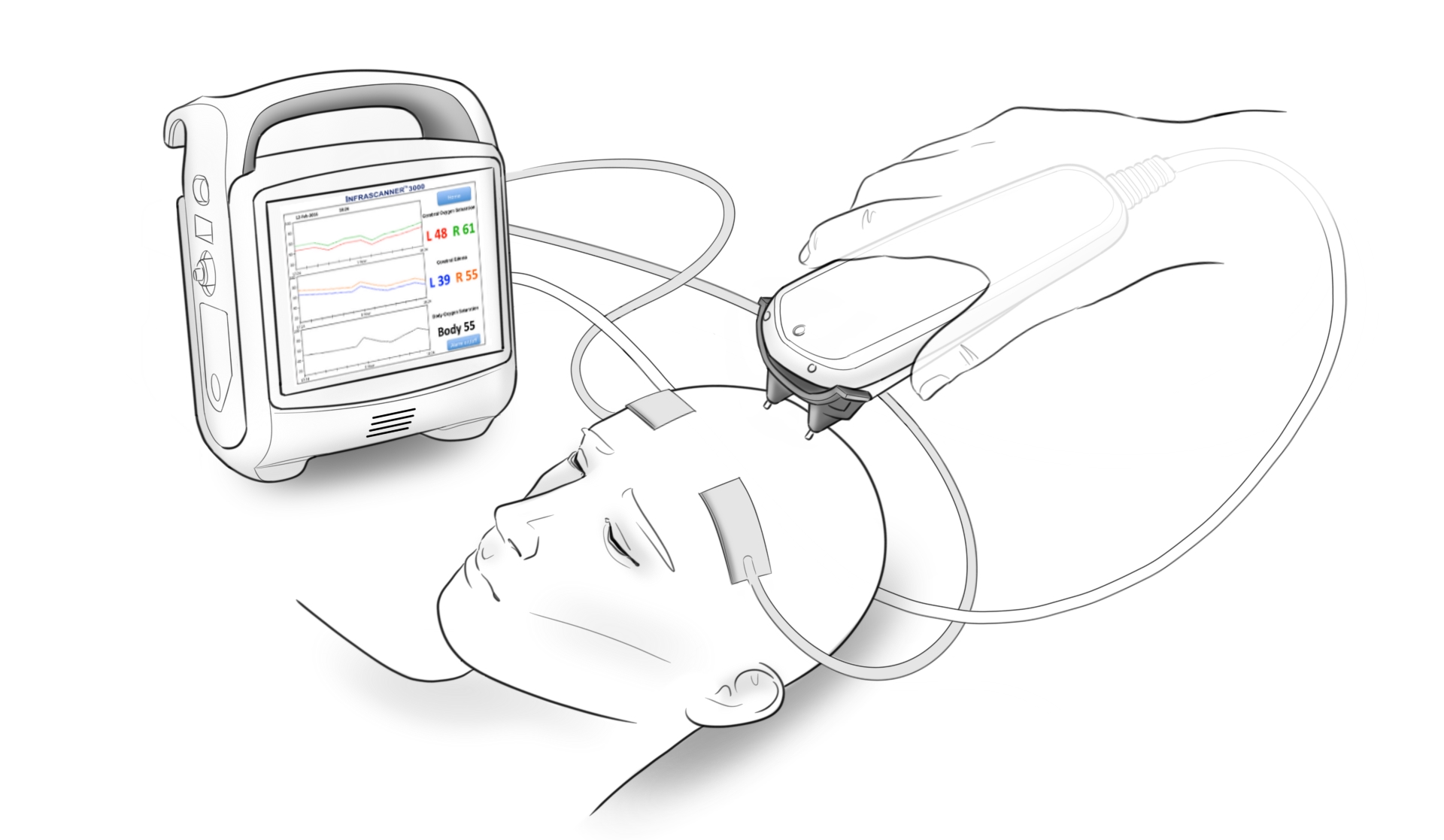
The patents cover the technologies to expand the capabilities of InfraScan's Infrascanner™, a hand-held, point-of-care device to detect bleeding in traumatic brain injury patients.
InfraScan has over 1,000 devices deployed in the US and internationally, including with some of the world's leading neurotrauma and trauma experts. InfraScan also licensed a variety of patents that apply to hemorrhagic and ischemic stroke detection and brain hemorrhage monitoring.
In the general population, an estimated 1.5 million individuals seek medical treatment for head trauma in the U.S. each year, and a total of 25 million individuals sustain head trauma annually worldwide. The Infrascanner has obtained FDA clearance, the CE mark, TGA, KFDA, and is now being marketed all over the world.
About InfraScan – InfraScan, Inc. is a medical device company that focuses on developing, commercializing and distributing portable brain scanner for head injury and stroke assessment based on near infrared (NIR) technologies. The company has received funding from the U.S. Army Medical Materiel Development Activity, U.S. Marine Corps, Office of Naval Research, BioAdvance, the Biotechnology Greenhouse of Southeastern Pennsylvania, Ben Franklin Technology Partners of Southeastern Pennsylvania, and from the Philadelphia Industrial Development Corporation. For more information visit http://www.infrascanner.com.
SOURCE InfraScan, Inc.

Share this article