New findings highlight growing interest in Cas-CLOVER editing for CHO cell engineering and biologics manufacturing.
LEXINGTON, Ky., April 15, 2026 /PRNewswire-PRWeb/ -- Recent research from scientists at Sanofi and Demeetra highlights growing interest in genome-editing architectures designed to deliver precise genetic modifications while potentially reducing off-target effects.
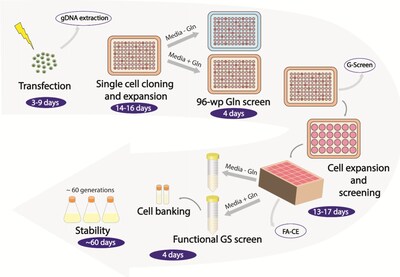
In the study Evaluation of gene editing in CHO cells using the Cas-CLOVER system, researchers evaluated Cas-CLOVER gene editing to knock out the glutamine synthetase locus in Chinese hamster ovary (CHO) cells. More than 100 clones were generated from multiple host cell lines, with complete knockout observed in 98 of them. The editing phase was completed in approximately six weeks, a timeline comparable to commonly reported CRISPR workflows.
The research evaluated 30 distinct serum-free CHO-K1 host cell lines, achieving functional glutamine synthetase knockouts across 28 of the 30 host backgrounds, demonstrating the scalability of the approach across diverse CHO platforms. Cas-CLOVER-mediated knockout clones and subclones remained stable over 63 days in culture, while editing efficiencies reached 72% using nucleofection, according to supplementary data reported in the study.
"At Sanofi, our biologics teams are exploring genome editing technologies to drive host cell engineering forward. Strengthening our cell line development capabilities requires optimizing CHO cells to unlock greater productivity and higher product quality, key pillars in building a scalable biologics pipeline that brings treatments to the patients who need them."
Researchers also reported monoclonal antibody titers increasing up to 14.4-fold compared with parental host cells, highlighting the potential productivity impact of genome engineering approaches in CHO cell systems.
"Biopharma teams are increasingly evaluating genome editing platforms that offer precise control over host cell engineering," said Jack Crawford, CEO of Demeetra. "As cell line development platforms evolve, the ability to engineer CHO for enhanced productivity and product quality becomes an important factor in supporting scalable biologics manufacturing."
The research also noted that Cas-CLOVER editing can generate larger insertion and deletion events than those typically produced by CRISPR-Cas9 editing. These larger indels may facilitate both gene knockouts and knock-ins by more effectively removing functional regions of target genes.
Chinese hamster ovary cells remain the dominant production platform for monoclonal antibodies and other biologics, and continued innovation in genome engineering technologies is helping developers refine cell line development strategies for improved productivity and consistency.
Demeetra develops genome-engineering and host-cell platforms to support commercial cell-line development and biologics manufacturing workflows. Follow Demeetra on LinkedIn.
Media Contact
Jack Crawford, Demeetra AgBio, 1 315-351-9115, [email protected], https://demeetra.com/
SOURCE Demeetra AgBio



Share this article