New VA brand by IXu: Female Condom Marketing Revolutionized; Launches in Europe
World's new VA® female condom brand launches online in Europe. The vawow.com site focuses on graphics of the condom in demonstration models. The new strategy highlights the pleasing visual aspects of its elegant and contoured fit as never before shown in female condom marketing.
PETOSKEY, Mich., March 8, 2019 /PRNewswire-PRWeb/ -- The designers of the VA® worn-of-women female condom brand announce its successful online launch in Europe.
The VA is sold exclusively online in Europe at the VA w.o.w website, vawow.com. CE-Mark approved for sale in Europe, the VA is expected to be available in the USA by the end of the year.
"Female condoms need to become more popular in Germany. We welcome the new VA to play a major role for women's sexual health," says Harriet Langanke from Germany's Charitable Foundation Sexuality and Health (GSSG) in Cologne, Germany.
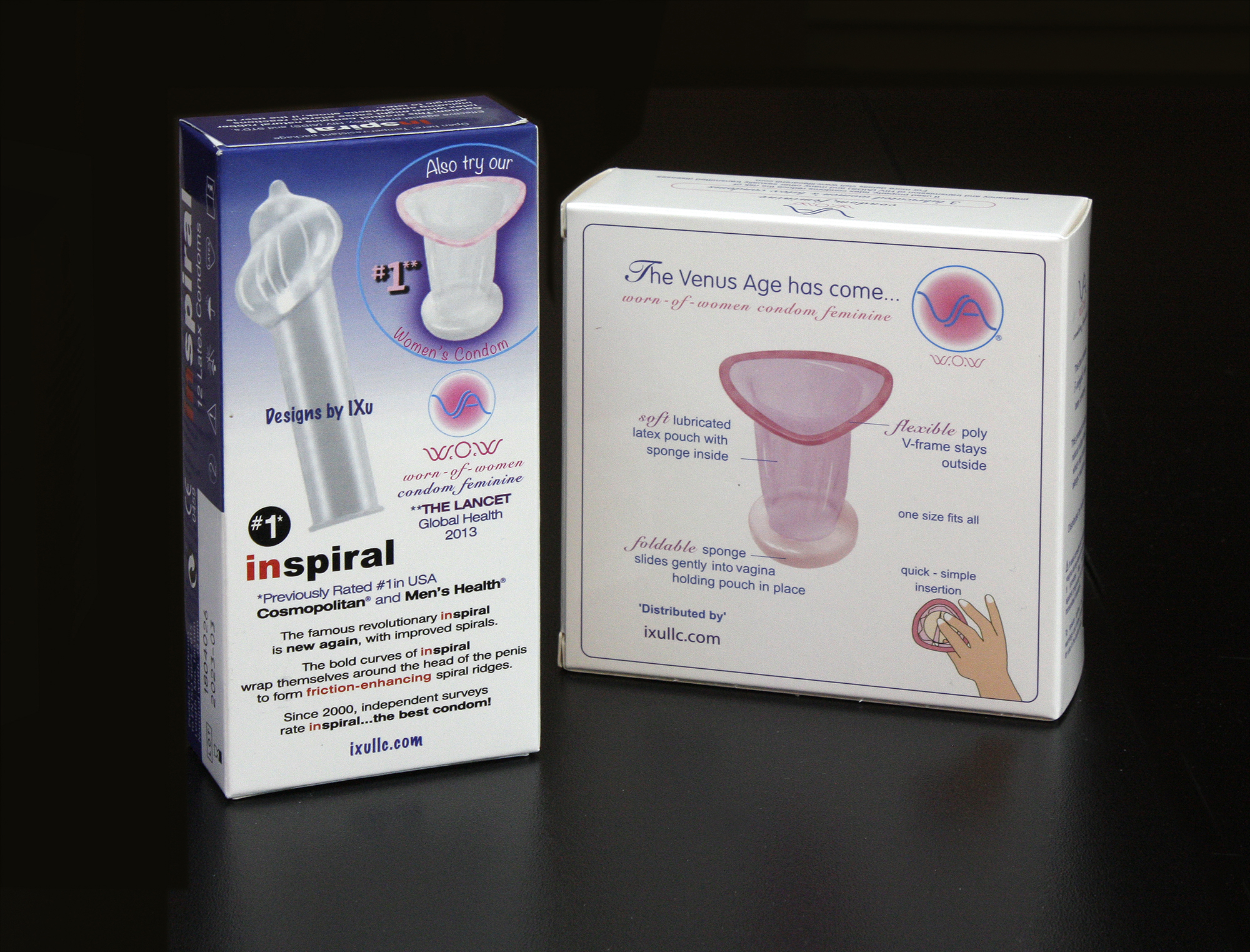
U.S. design firm IXu said the popularity of the VA in European, Brazilian and African test markets prompted their manufacturing contract with major global condom maker HLL Lifecare Ltd. of India.
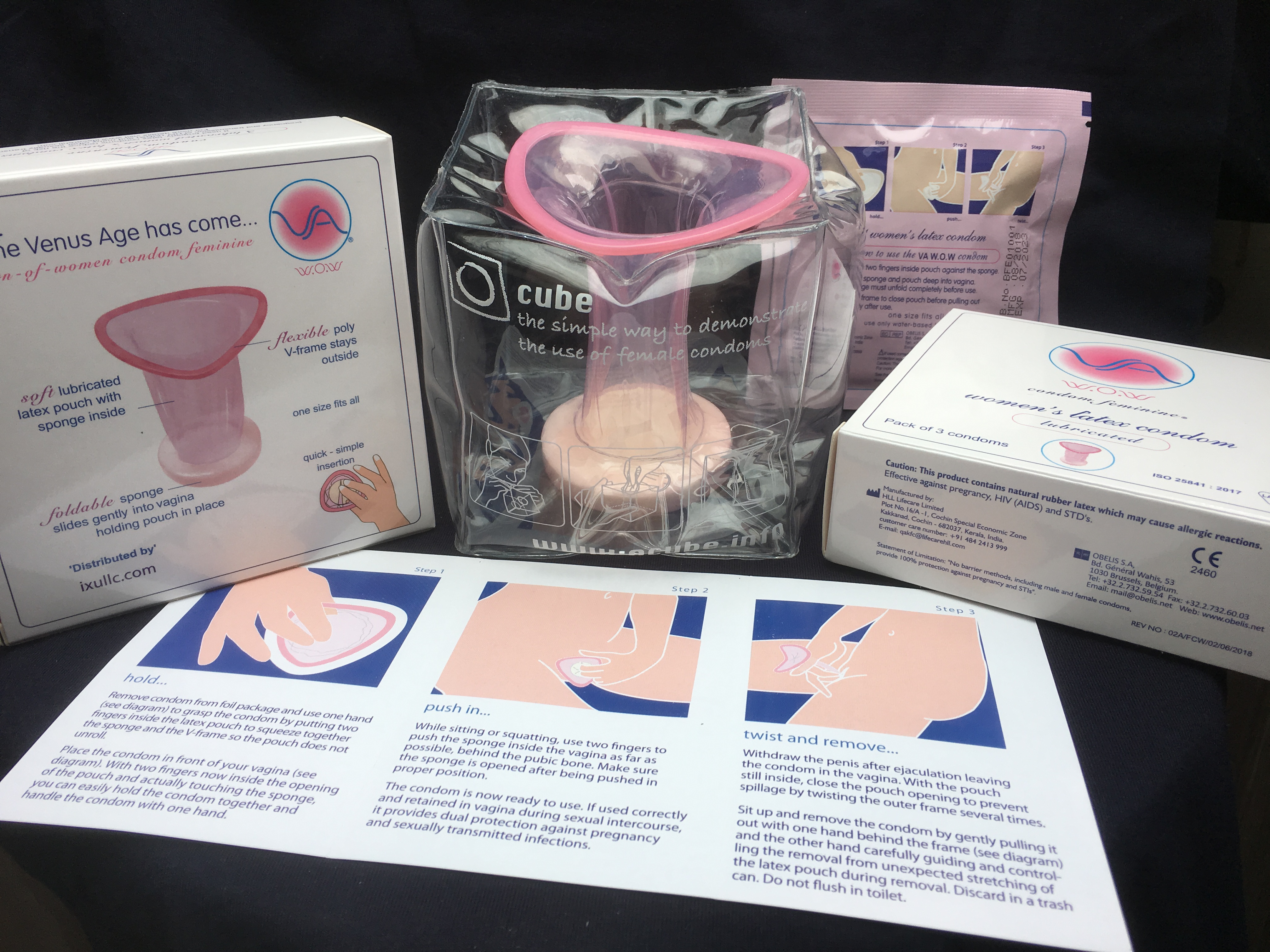
The VA obtained the highest efficacy rating in a comparison of all four European CE-Mark approved female condoms in an independent clinical study published in the British Lancet Medical Journal. And the VA achieved a 97% satisfaction rating in a user survey.
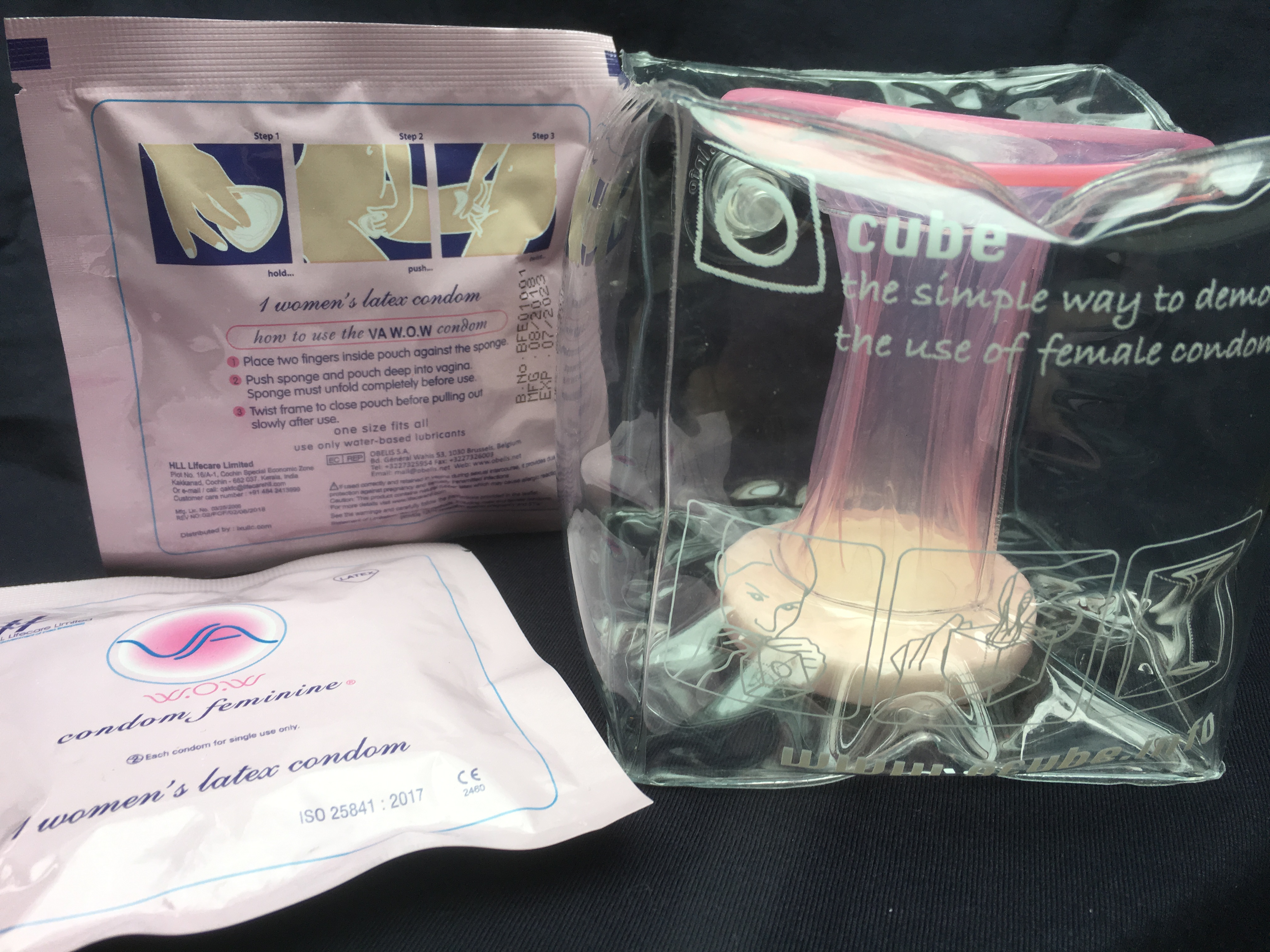
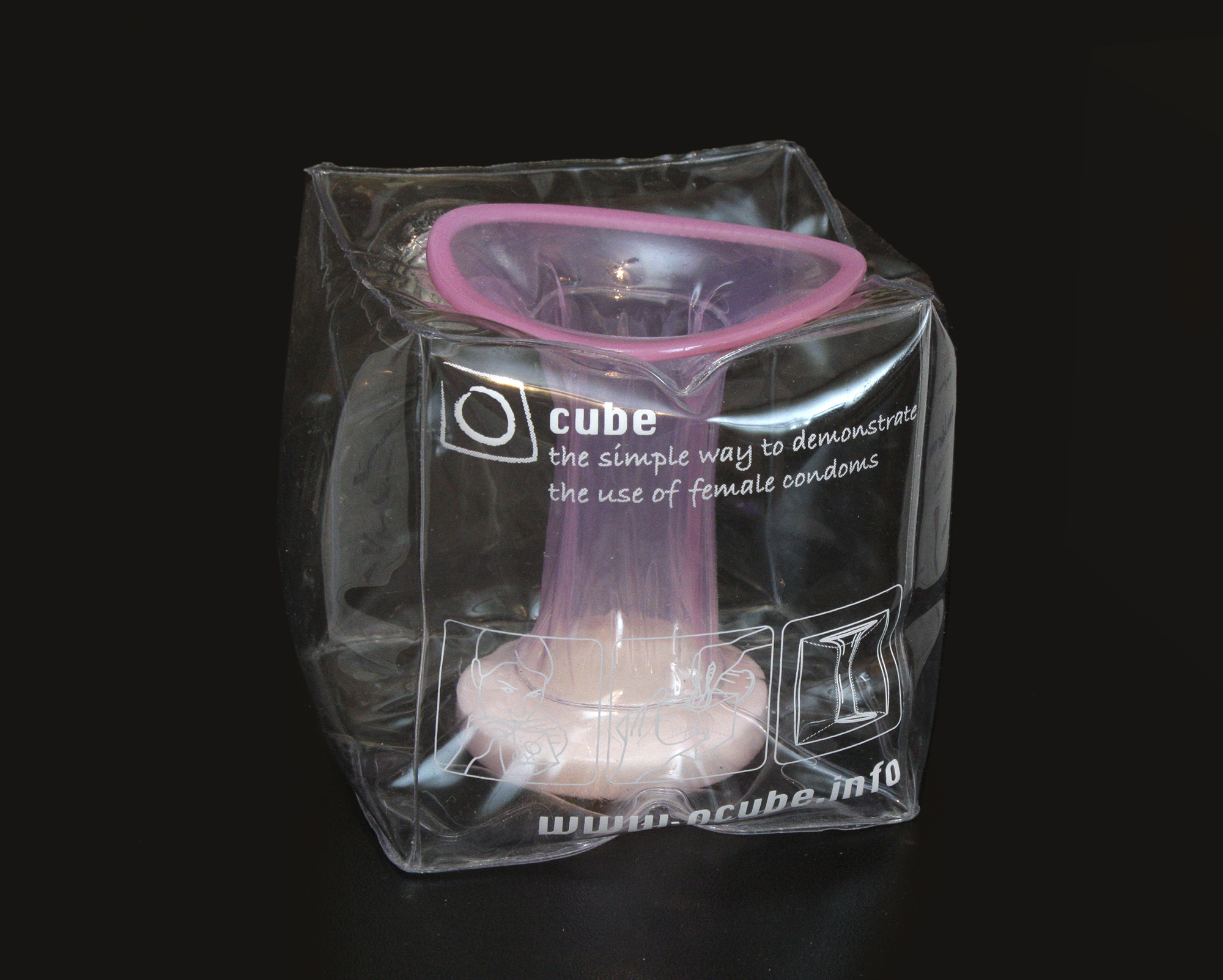
IXu CEO Brian Osterberg states, "We're pleased to finally offer global consumers the sensual-looking, body-fitting female condom that's been missing from the market. In test markets, the VA easily became more popular than the older-known brand, the brand recently dropped from the U.S. retail market by its own maker. The new VA looks nothing like the older brand. The pink latex VA is extremely sleek and feminine, almost fashionable in appearance and fit, as seen in the demonstration models. We have simply changed the way female condoms are designed and marketed to women."
The VA registered a successful two-year trial market at all Portuguese public health clinics. Distributed by the Clinifar company of Lisbon, after one year, the VA was selected as the only brand to be offered in clinics, due entirely to consumer response.
"Clinifar is pleased to once again offer the VA condom, as the Portuguese Health Ministry had success with the popularity of the VA in nationwide clinics in a prior trial market," states Monica Costa, Manager, Clinifar, Lisbon, Portugal.
IXu states the VA proved more than the functional equivalent of the only US FDA-approved female condom, surpassing it in efficacy ratings as published in the Lancet medical journal.
HLL Lifecare Ltd. is the exclusive contract-manufacturer of the VA for IXu LLC.
"The demand for simple, effective and inexpensive latex female condoms has been proven with our VA condom, and should increase greatly in the years to come with proven popularity," said Mr. T. Rajasekar, Director of Marketing, HLL, "Our launching in Europe allows it to become a global game-changer for safe sex. HLL is delighted to partner exclusively with IXu for the manufacture of VA W.o.W condoms for women."
IXu will expand the VA line this year with a VA Vibe model featuring miniature vibrating accessories attached to a standard VA condom.
Contact:
Brian Osterberg, CEO
IXu LLC
Petoskey, MI 49770 USA
USA phone: 001 231 242 4440
vawow.com
ixullc.com
[email protected]
[email protected]
SOURCE IXu
Share this article