Fast and Efficient: Why Utilizing Electronic Diaries can be more Cost Effective than Paper Ones
WILMINGTON, Del., Dec. 20, 2017 /PRNewswire-iReach/ -- Subject diary is an integral part of clinical research. According to the FDA statistics, patient diaries are used in more than 25% of clinical trials. For many Sponsors, the choice of a recording method is ambiguous since both types of diaries have their advantages: paper diaries are traditionally considered to be more conventional and cheaper, while their electronic counterparts are often praised for easy data transfer, convenient interface, and notifications enabling timely reporting.
This article shows how electronic collection of patient reported outcomes which determine primary study endpoints can shorten terms and save costs of a clinical trial versus conservative recording methods.
In 2016, Smooth Drug Development developed and validated its own ePRO solution. The Smooth application for mobile devices (e.g. tablets and smartphones) enables to accumulate data, send it to the data-centers, and make it available for medical personnel and clinical monitors in real time.
In 2016-2017, the Smooth Drug Development team had an opportunity to compare the efficacy of electronic and conservative methods of data collection in two studies of analogue products, with similar designs in the homogenous population of patients. In the first study, the researchers employed paper diaries (research I, blue graphs below). In the second one, the investigators offered patients to use Smooth electronic diaries (research II, red graphs). All data was blinded and processed in a way that enables to conduct a comparative analysis.
In the result of the comparative analysis, specialists of Smooth Drug Development came to important conclusions, which bring in new arguments on why ePROs can be more effective than paper ones.
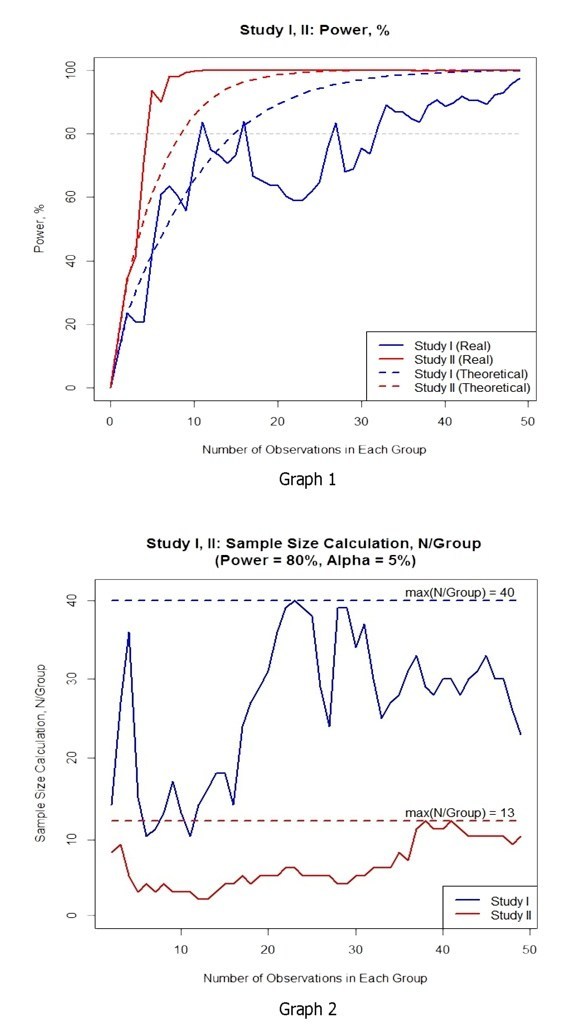
1. The study with utilization of ePRO solution demonstrated significantly lower variance. This factor contributed to faster achievement of the statistical power of a test in the clinical trial.
The first graph shows the comparison of theoretical (calculated on all observations) and real (calculated on observations at each time-point) test power in each study. In the graph, one can observe that the real power increase and achievement of the required 80% in study II progresses faster than in the theoretical one. This effect is explained by more homogenous data. The results of study I show higher data variation and slower achievement of the required minimum.
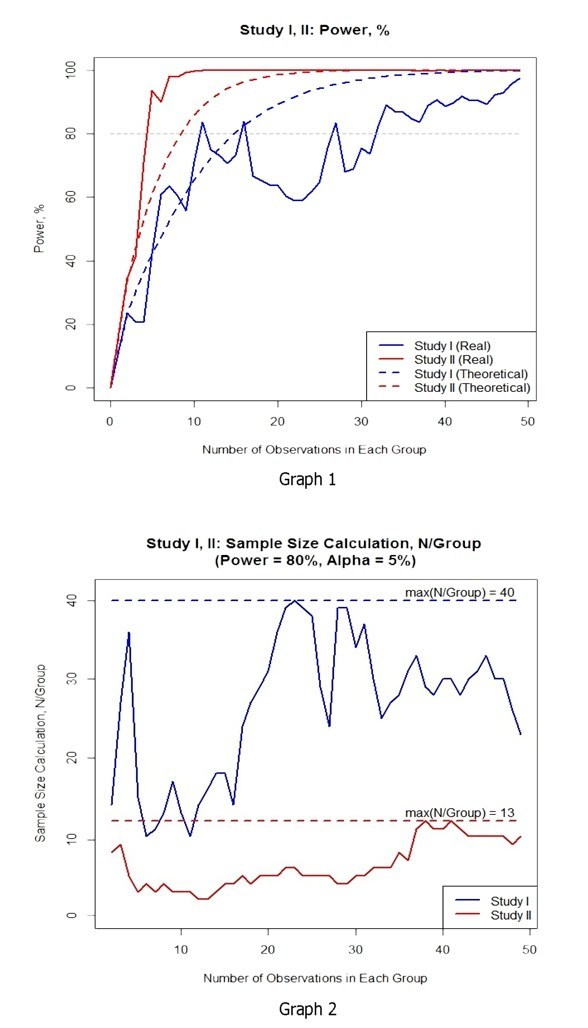
2. In the trial with electronic diaries, the necessary test power of 80% was achieved in a considerably smaller patient sample size. This conclusion implicates that it is possible to achieve the results of the clinical trial engaging fewer patients than in trials with paper diaries.
Graph 2 demonstrates interdependency between the sample size necessary for achieving the test power of 80%, and the number of enrolled patients. In the second graph, the required power is arranged in a narrower corridor, and the maximum value of the sample size is 67,5% lower compared to the same criterion in graph 1.
Possible reasons for such a difference:
- Paper diaries don't give an opportunity to control its completion time exactly. Therefore, we assume that a certain part of diaries in the study was filled in by the patient late (e.g., prior to their next study visit), or even partly omitted. Inaccurate and incomplete data raise variance.
- The sample sizes were calculated based on the published data of previously conducted studies. Since in most such studies paper diaries were used, coefficients of variation take into account this feature connected to the use of paper diaries, which is described above.
This example demonstrates that clinical trials with primary endpoint assessment based on the patient diary data can benefit from using electronic systems that lower data variance and help to achieve sufficient test power at a relatively smaller population of subjects.
We analyzed how these advantages can be converted to a value and assumed that usage of an adaptive study design enables to stop the study pre-term based on positive results of an interim analysis, thus decreasing the cost and timelines of its conduct.
Source: © 2017, Smooth Drug Development
Media Contact: Ivan Grek, Smooth Drug Development, +12024152208, [email protected]
News distributed by PR Newswire iReach: https://ireach.prnewswire.com
SOURCE Smooth Drug Development
iReach LastName, iReachCompany, 111-222-3333, [email protected]
Share this article