FDA Clearance of the SkinPen® Precision Microneedling Device Makes it the ONLY Safe and Legally Marketed Microneedling System in the U.S.
ADDISON, Texas (PRWEB) March 06, 2018 -- Bellus Medical, a leader in medical aesthetics products and devices, has been granted clearance and marketing authorization by the FDA for its medical grade microneedling device, SkinPen® Precision, used exclusively by healthcare professionals. This is the first and only microneedling device in the industry to receive this important designation from the U.S. Food and Drug Administration (FDA), and sets a new standard as the only recognized Class II microneedling device available.
"FDA clearance for SkinPen Precision demonstrates our unwavering commitment to the safety, quality and excellence needed to elevate the standards in the microneedling industry," said Joe Proctor, Bellus Medical President and CEO. "With this recognition by the highest organization in the U.S., the FDA, healthcare providers and consumers know they can trust SkinPen Precision and Bellus Medical to create, develop and manufacture the safest and most cutting-edge solutions on the market."
Proactive Clinical Evaluation and Proven Results
To receive this first-in-class FDA designation, Bellus Medical proactively worked through a rigorous three-year evaluation process to meet more than 90 validated requirements for the microneedling device, charger base and proprietary cartridge, including extensive biocompatibility testing to ensure none of the materials are harmful to patients' skin cells.
"Our evaluation process with the FDA resulted in a new draft guidance issued by the FDA, which includes regulatory requirements for microneedling devices," said Jennifer Block, Bellus Medical Director of Quality & Regulatory. "Our steadfast focus on healthcare provider and patient safety resulted in a new classification for microneedling devices. Bellus Medical's SkinPen Precision is truly leading the industry as the only microneedling system on the market meeting the FDA's new standards."
Healthcare Providers and Patients Choose SkinPen® Precision
Bellus Medical collaborated with some of the world's leading experts in the medical aesthetics industry, dermatology and plastic surgery to help identify the safety risks associated with microneedling devices. This feedback, along with the SkinPen Precision clinical trials, helped Bellus Medical implement measures to prevent risks to healthcare providers and consumers.
Video Testimonials
- Watch Dr. Angobaldo discuss why he chooses SkinPen for his patients: https://youtu.be/4J7ZIhM2O3E
- Hear why Kimberly loves SkinPen for herself and recommends it to others: https://youtu.be/qnQ68EDMGBA
Healthcare providers and consumers can experience highly effective meaningful benefits offered only by SkinPen Precision, including:
- Little to no downtime
- Effective for all skin types, light to dark
- Comfortable experience
- Short procedure time
- Ideal any time of the year
- Real results from a safe, natural and minimally invasive procedure
- Clinically-proven, safe and effective
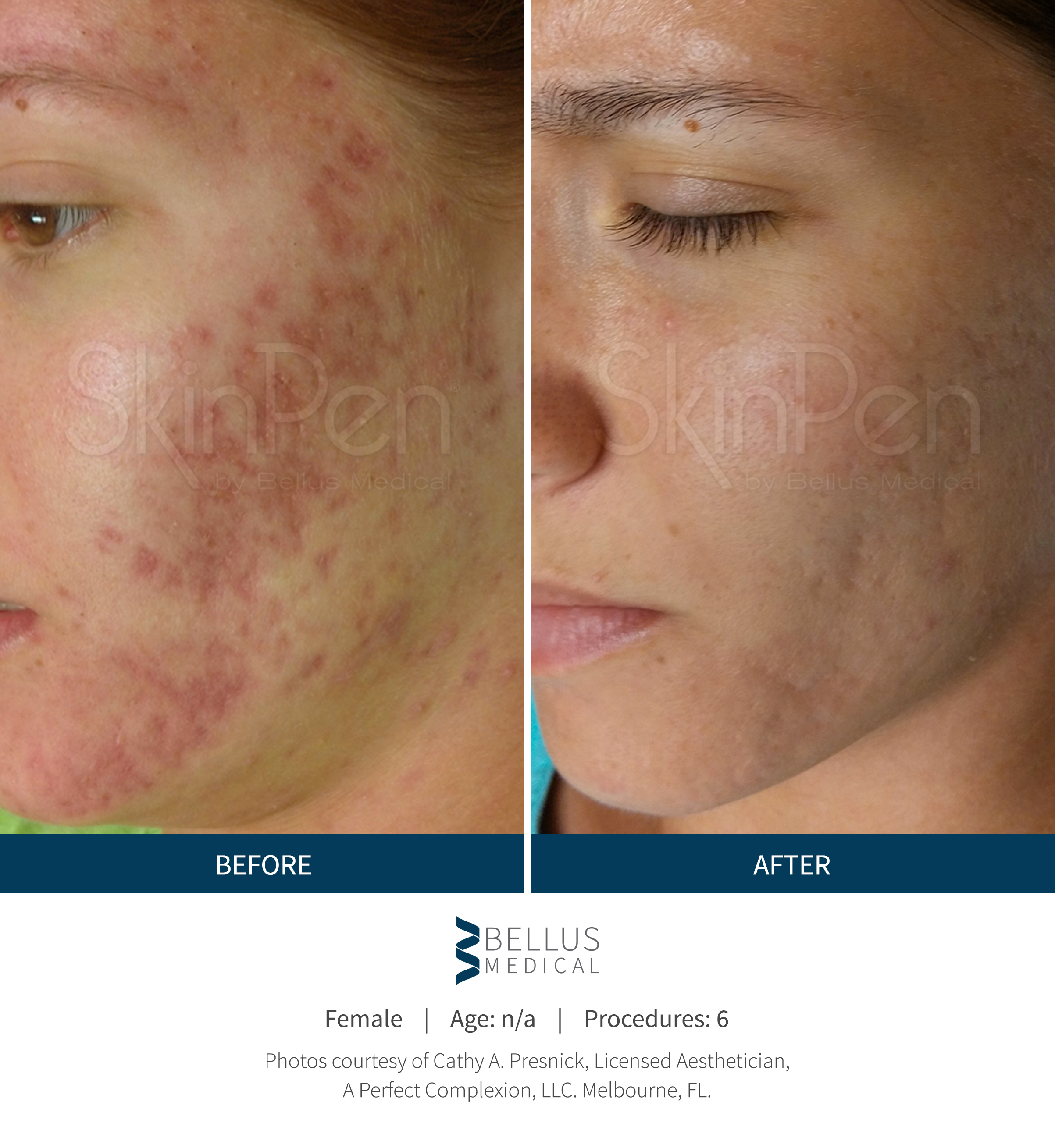
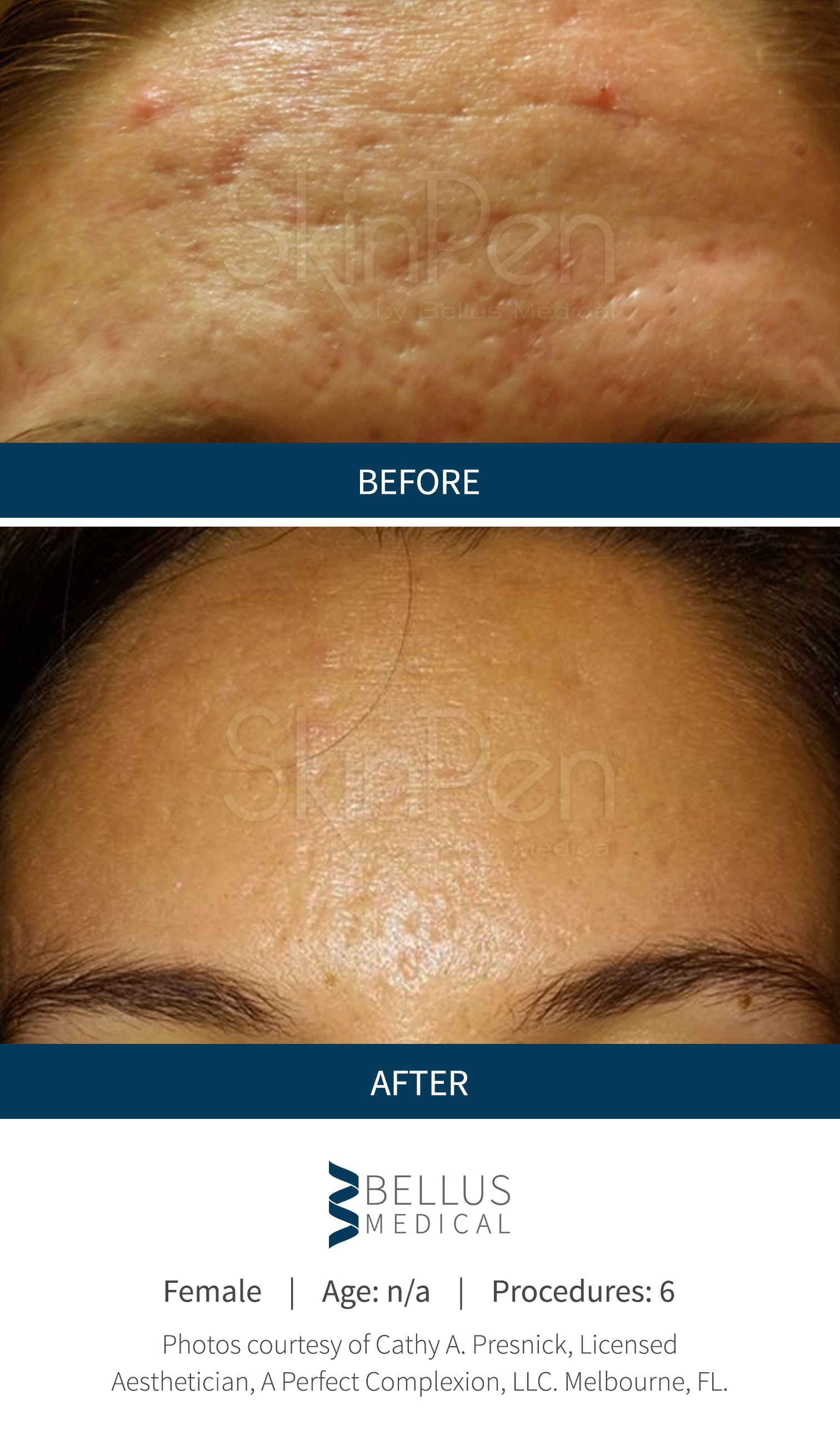
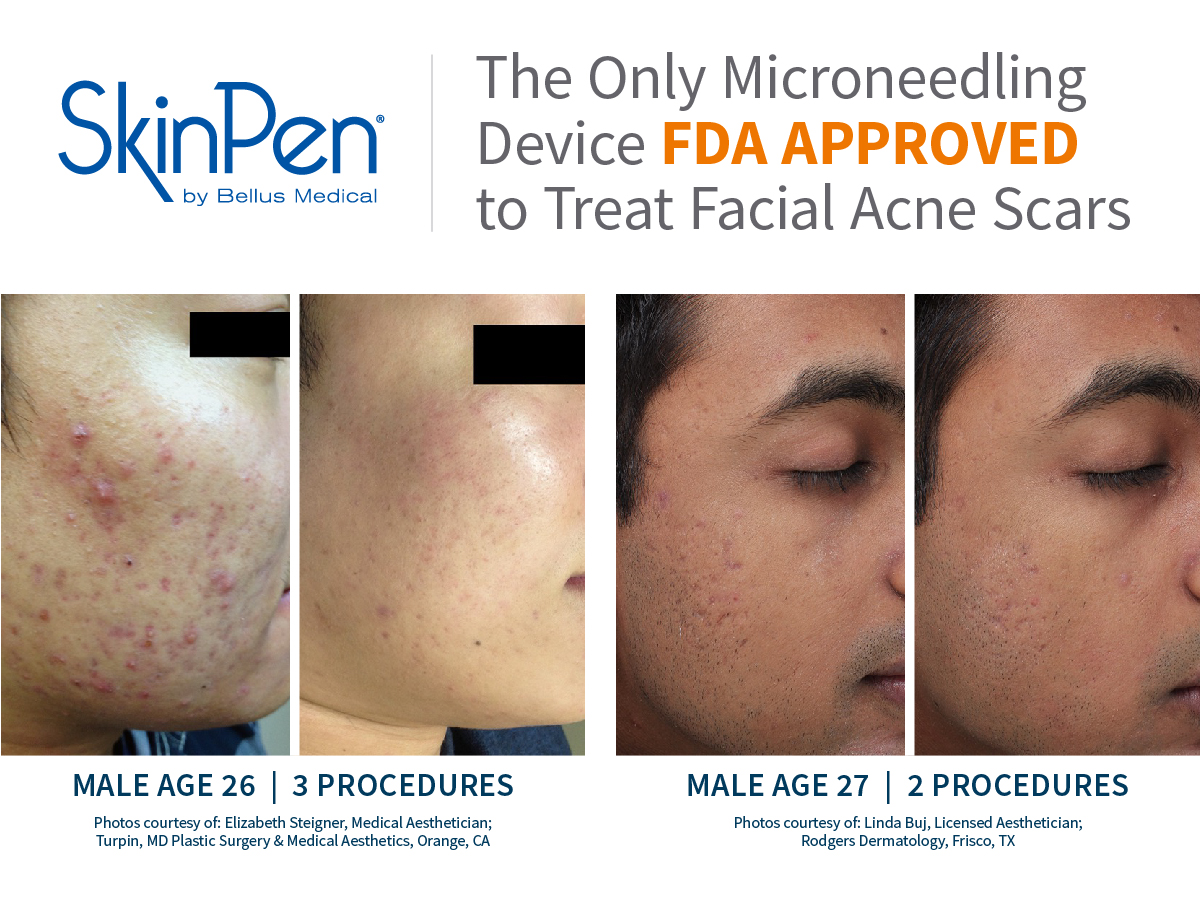
These SkinPen Precision benefits are backed by findings in Bellus Medical's clinical trials, as well as satisfaction reported by the trial's subjects. Patients increasingly reported improvement in the appearance of their acne scars, and at the end of 6 months, 90% reported an improvement in the appearance of their acne scars. Further, 90% said they would recommend SkinPen Precision treatment.
The SkinPen Precision Microneedling System has been evaluated and is appropriate for all Fitzpatrick skin types, light to dark. The results from the SkinPen Precision clinical trial showed treatments with the SkinPen Precision system improve the appearance of facial acne scars.
For complete clinical trial summary and information on how to use the SkinPen Precision microneedling device, visit skinpen.com.
Bellus Medical Always Puts Safety First
FDA Clearance follows Bellus Medical's other recent safety and quality milestones including the CE Mark registration for SkinPen Precision Microneedling system, which allows Bellus to sell the SkinPen Precision microneedling system commercially to physicians within the European Union. In addition, Bellus Medical is the first microneedling device manufacture to receive ISO 13485:2016 Certification, reinforcing the company's commitment to providing the safest, highest-quality and most cutting-edge devices and products on the market.
SkinPen® has more than 90 validated requirements for the pen, charger base and cartridge, and passed extensive biocompatibility testing. This includes testing for irritation and cytotoxicity, and sensitization ensuring none of the materials used to manufacture the SkinPen Precision system are harmful to the patient's skin cells. SkinPen Precision is also the only microneedling device with software validation to guarantee performance of its smart technology.
About Bellus Medical
Bellus Medical, headquartered in Addison, TX, is an industry leader in medical aesthetics with an unmatched reputation of providing the highest levels of safe and effective products, and the most innovative technology on the market. Founded in 2012, Bellus quickly became an industry pioneer with the introduction of its flagship product SkinPen®, a state-of-the-art microneedling device, which is now the only FDA Cleared medical grade microneedling device on the market. Bellus' product portfolio continually expands with unique and differentiated products developed to enhance the patient experience and represent a significant business opportunity for medical practices. Built upon three guiding principles - technology, education and customer service - Bellus is much more than a supplier to aesthetic practices, it is a trusted business partner. To learn more about Bellus Medical, visit BellusMedical.com.
Intended Use:
The SkinPen Precision system is a microneedling device and accessories intended to be used as a treatment to improve the appearance of facial acne scars in adults aged 22 years or older.
Media Contact
Megan Lozito
megan(at)beanstalkdigital.com
214-924-8927
Megan Lozito, Bellus Medical, LLC, http://www.skinpen.com, +1 214-924-8927, [email protected]





Share this article