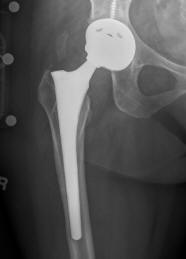
US Drug Watchdog Now Expands Their National All Metal Hip Implant Victims Initiative With The Goal Being Identifying More Recipients With A Premature Failure
(PRWEB) August 22, 2013 -- The US Drug Watchdog intends to dramatically increase their efforts to identify people, who are recipients of what are called metal on metal hip implants, if their device has failed, or is in the early stages of a premature failure. The group is the best branded source on the Internet for people who have a all metal hip implant, and a blood test designed to indicate a premature failure. However, they now fear without a more robust effort, potentially hundreds of thousands of US citizens, who have these specific types of hip implant devices will get left on the curb, stuck with huge medical bills related to the replacement costs associated with a new hip implant.
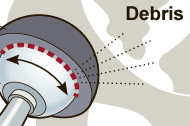
The US Drug Watchdog says, "We are retooling our efforts to identify more recipients of a metal on metal hip implant, because in our opinion only a fraction of recipients who have a failure, or symptoms of a premature failure have been identified to the courts, that are handling these various premature failure lawsuits. In addition to making suggestions about how to test for a premature failure, we will start including information related to deadlines, in which a recipient must be identified to the appropriate court, if they have any hope of being compensated by the medical device company, that produced their potentially defective metal on metal hip implant." For more information recipients of a metal on metal hip implant are urged to contact the US Drug Watchdog at 866-714-6466.
The US Drug Watchdog says, "Litigation involving metal on metal hip implants prematurely failing include the following manufacturers:"
- DePuy Pinnacle Case Number U.S. District Court for the Northern District of Texas MDL#2244
- DePuy ASR Case Number Ohio Southern Federal District Court Case Number MDL No. 2197AAJ
- Stryker Rejuvenate Hip Implant Stem Failure Teleseminar: Metallosis with No Metal on Metal Articulation will address the issues surrounding the latest hip replacement systems recall. On July 6, 2012 Stryker issued a voluntary recall of its Rejuvenate and ABG II modular-neck stems in the United States
- Biomet M2a Magnum Metal-on-Metal Hip Replacement U.S. District Court for the Northern District of California. (Case No. 3:12-cv-3476) filed on July 2nd 2012
- Zimmer Durom Cup Product Liability mass tort litigation, MDL NO. 2158 (SDW), which is pending in the federal District Court in New Jersey (time frame 2006-July 2008)
- Wright Medical Technology, Inc., Conserve Hip Implant Products Liability Litigation ("MDL No. 2329"). This is now pending in the U.S. District Court for the Northern District of Georgia.
The US Drug Watchdog says, "One of the biggest problems we have with respect to our failed medical device or recalled drug initiative work is the average US consumer never hears about a drug recall or failures involving medical devices like metal-on-metal hip implants. If you have a friend, or loved one who is a recipient of a metal-on metal hip implant that has already failed or is showing symptoms of a failure, or a recipient is concerned about a premature failure, please call us anytime at 866-714-6466. We want to make certain all metal-on-metal hip implant recipients get meaningful help, and assistance." http://USDrugWatchdog.com
Important note from the US Drug Watchdog: "The link to the December 27th 2011 New York Times article titled,"The High Cost of Failing Artificial Hips is no longer active. However we believe the article is extremely relevant, so we encourage interested parties to Google the article by title, and date. Further according to the New York Times on May 16th 2013 Johnson & Johnson will soon be getting out of the all metal hip implant business." http://www.nytimes.com/2013/05/17/business/jj-is-phasing-out-metal-replacement-hips.html
M Thomas Martin, Americas Watchdog, 866-714-6466, [email protected]

Share this article